Enthalpy Of Formation Of Liquid Water
News Leon
Apr 02, 2025 · 6 min read
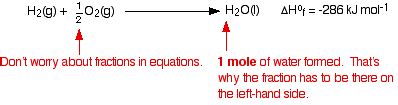
Table of Contents
Enthalpy of Formation of Liquid Water: A Deep Dive
The enthalpy of formation of liquid water, denoted as ΔfH°(H₂O, l), is a fundamental thermodynamic quantity with significant implications across various scientific disciplines. Understanding its value and the processes involved in its determination is crucial for comprehending chemical reactions, energy transfers, and the behavior of matter. This article will delve into the intricacies of this thermodynamic property, exploring its definition, calculation methods, applications, and the factors influencing its value.
Understanding Enthalpy of Formation
Before focusing specifically on water, let's establish a clear understanding of enthalpy of formation. Enthalpy (H) is a thermodynamic state function representing the total heat content of a system at constant pressure. It's a crucial concept for understanding energy changes in chemical reactions. The enthalpy of formation (ΔfH°) is the change in enthalpy that occurs when one mole of a compound is formed from its constituent elements in their standard states (usually at 298.15 K and 1 atm pressure). The standard state signifies the most stable form of the element under these conditions. For example, the standard state of oxygen is O₂(g), not O(g) or O₃(g).
A negative enthalpy of formation indicates an exothermic process, meaning the formation of the compound releases heat to the surroundings. Conversely, a positive enthalpy of formation signifies an endothermic process, where heat is absorbed from the surroundings during compound formation.
Determining the Enthalpy of Formation of Liquid Water
The enthalpy of formation of liquid water is experimentally determined using calorimetry. Several methods exist, each with its own advantages and limitations:
1. Direct Combustion Calorimetry:
This method involves directly burning hydrogen gas (H₂) in oxygen gas (O₂) under controlled conditions within a bomb calorimeter. The heat released during the reaction is measured precisely, and this value, along with the stoichiometry of the reaction, is used to calculate the enthalpy of formation. The reaction is represented as:
2H₂(g) + O₂(g) → 2H₂O(l)
The heat released is directly proportional to the enthalpy change of the reaction. Careful calibration and consideration of heat losses are essential for obtaining accurate results. This method is widely used due to its relative simplicity and accuracy.
2. Hess's Law:
When direct measurement is difficult or impractical, Hess's Law provides an indirect approach. This law states that the enthalpy change of a reaction is independent of the pathway taken. Therefore, the enthalpy of formation can be calculated using a series of known enthalpy changes for other reactions that sum up to the desired formation reaction. This often involves using standard enthalpies of formation for other compounds, readily available in thermodynamic tables. For instance, one could utilize the enthalpies of formation of relevant oxides and then use Hess’s Law to calculate the enthalpy of formation of liquid water. This method requires precise data for the intermediate reactions.
3. Computational Methods:
With advancements in computational chemistry, theoretical calculations can provide estimates of the enthalpy of formation. Sophisticated quantum mechanical methods, such as Density Functional Theory (DFT), are employed to model the electronic structure and energy of molecules. These methods can provide insights into the bonding interactions within the water molecule, allowing for an estimation of its formation enthalpy. While highly sophisticated, the accuracy of these computations depends on the level of theory and the approximations used.
The Value of ΔfH°(H₂O, l)
The accepted standard enthalpy of formation for liquid water at 298.15 K and 1 atm is approximately -285.8 kJ/mol. The negative sign confirms that the formation of liquid water from its constituent elements is a highly exothermic process. This large negative value reflects the strong hydrogen bonding present in liquid water, a significant factor contributing to its stability and unique properties.
Factors Influencing the Enthalpy of Formation
Several factors can influence the measured enthalpy of formation of liquid water, including:
-
Temperature: The enthalpy of formation is temperature-dependent. While the standard value is given at 298.15 K, variations from this temperature will result in different enthalpy values. These temperature dependencies are often expressed using empirical equations or heat capacity data.
-
Pressure: Pressure influences the enthalpy of formation, although the effect is generally less significant than temperature variations, especially at pressures close to standard atmospheric pressure. At very high pressures, the compressibility of the reactants and products needs to be considered.
-
Isotope effects: The isotopic composition of hydrogen and oxygen can slightly affect the enthalpy of formation. For instance, using deuterium (²H) instead of protium (¹H) will lead to a slightly different enthalpy value due to the difference in mass and vibrational frequencies.
-
Experimental Errors: Experimental errors inherent in calorimetric measurements can introduce uncertainty into the determined value. Careful calibration, precise measurements, and accounting for heat losses are crucial for minimizing these errors.
Applications of Enthalpy of Formation Data
The enthalpy of formation of liquid water, along with other thermodynamic data, has widespread applications in various fields:
-
Chemical Engineering: It’s crucial for designing and optimizing chemical processes, particularly those involving water as a reactant or product. For instance, it helps in calculating the energy balance and efficiency of industrial processes such as steam generation or water electrolysis.
-
Environmental Science: Understanding the enthalpy of formation of water is important for analyzing environmental processes such as rainfall formation, the energy involved in hydrological cycles, and the impact of climate change.
-
Geochemistry: It's essential for modeling geochemical processes, such as mineral weathering and the formation of hydrothermal systems, where water plays a critical role.
-
Thermochemistry: It serves as a fundamental parameter in thermodynamic calculations, allowing for the prediction of equilibrium constants and reaction spontaneity.
-
Materials Science: The value aids in understanding the stability and reactivity of materials in aqueous environments. It's valuable in assessing corrosion processes and the behavior of materials under different hydration conditions.
Conclusion
The enthalpy of formation of liquid water is a cornerstone parameter in thermodynamics, with significant implications across numerous scientific disciplines. Its determination through various experimental and computational methods provides crucial insights into the energetics of water formation and its role in various chemical and physical processes. The negative value reflects the high stability of liquid water under standard conditions, a consequence of its strong hydrogen bonding. Understanding this thermodynamic property is essential for advancements in fields ranging from chemical engineering to environmental science and materials science, highlighting its fundamental importance in our comprehension of the natural world. Further research continues to refine the accuracy of its measurement and expand our understanding of its nuanced dependence on various physical parameters. The value and its applications are undeniably far-reaching, impacting numerous aspects of our scientific and technological endeavors.
Latest Posts
Latest Posts
-
Weight Of An Apple In Grams
Apr 03, 2025
-
Find The Matrix X Such That Ax B
Apr 03, 2025
-
Distance Between Two Parallel Planes Formula
Apr 03, 2025
-
What Particle Determines The Identity Of An Element
Apr 03, 2025
-
Which Statement Is True About Bacteria
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Enthalpy Of Formation Of Liquid Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
