Electron Dot Structure For Magnesium Oxide
News Leon
Apr 04, 2025 · 5 min read
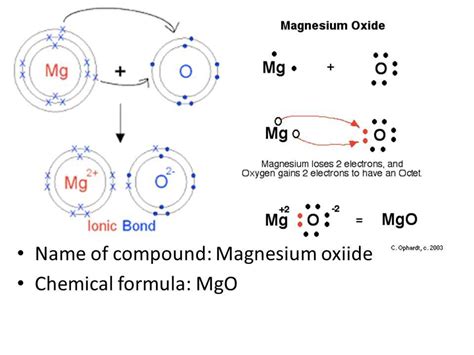
Table of Contents
Unveiling the Electron Dot Structure of Magnesium Oxide: A Deep Dive
Magnesium oxide (MgO), a ubiquitous compound with diverse applications, offers a fascinating case study for understanding chemical bonding through the lens of electron dot structures. This article delves deep into the intricacies of MgO's electron dot structure, exploring its formation, implications, and relevance in various scientific domains. We'll cover the basics, delve into the nuances, and equip you with a solid understanding of this fundamental concept in chemistry.
Understanding Electron Dot Structures: A Quick Recap
Before we dive into the specifics of magnesium oxide, let's refresh our understanding of electron dot structures, also known as Lewis dot structures. These diagrams visually represent the valence electrons – the electrons in the outermost shell of an atom – which are primarily responsible for chemical bonding. Each valence electron is depicted as a dot surrounding the element's symbol.
Why are electron dot structures important? They provide a simplified, yet powerful way to:
- Visualize valence electrons: This allows us to readily understand an atom's bonding capacity.
- Predict chemical bonding: We can see how atoms will share or transfer electrons to achieve a stable electron configuration (usually an octet).
- Represent molecular structures: For molecules, the dots show how atoms are connected and the distribution of electrons in bonds.
The Individual Atoms: Magnesium and Oxygen
To construct the electron dot structure for MgO, we must first examine the individual atoms involved: magnesium (Mg) and oxygen (O).
Magnesium (Mg)
Magnesium is an alkaline earth metal located in Group 2 of the periodic table. This means it has two valence electrons. Therefore, its electron dot structure is represented as:
Mg: .
Oxygen (O)
Oxygen is a nonmetal in Group 16 of the periodic table. It has six valence electrons. Its electron dot structure is:
:Ö:
Forming the Magnesium Oxide (MgO) Electron Dot Structure: The Ionic Bond
Magnesium oxide is an ionic compound, meaning it's formed through the electrostatic attraction between oppositely charged ions. Magnesium readily loses its two valence electrons to achieve a stable electron configuration resembling that of the noble gas neon (Ne). Oxygen, on the other hand, readily gains two electrons to achieve a stable electron configuration resembling that of the noble gas neon as well.
This electron transfer results in the formation of:
-
A magnesium ion (Mg²⁺): Magnesium loses two electrons, becoming a positively charged cation with a complete outer electron shell. Its electron dot structure is simply:
Mg²⁺ -
An oxide ion (O²⁻): Oxygen gains two electrons, becoming a negatively charged anion with a complete outer electron shell (octet). Its electron dot structure is:
[:Ö:]²⁻
The strong electrostatic attraction between the positively charged magnesium ion and the negatively charged oxide ion forms the ionic bond in MgO.
Therefore, the electron dot structure for magnesium oxide doesn't show covalent bonds (shared electrons) but instead represents the ionic interaction:
Mg²⁺ [:Ö:]²⁻
This simple representation reflects the transfer of electrons and the resulting charges on the ions.
The Crystal Lattice Structure of MgO: Beyond the Simple Dot Structure
While the simple electron dot structure illustrates the electron transfer, it doesn't fully capture the three-dimensional arrangement of ions in magnesium oxide. MgO exists as a crystalline solid with a face-centered cubic (FCC) structure. In this structure, each magnesium ion (Mg²⁺) is surrounded by six oxide ions (O²⁻), and each oxide ion is surrounded by six magnesium ions. This arrangement maximizes electrostatic attraction and minimizes repulsion, resulting in a highly stable structure. Understanding the crystal lattice is crucial for comprehending the macroscopic properties of MgO, such as its high melting point and hardness.
Properties of Magnesium Oxide (MgO) and Their Relation to Electron Dot Structure
The electron dot structure, while seemingly simple, underpins many of MgO's important properties:
-
High Melting Point: The strong electrostatic attraction between the Mg²⁺ and O²⁻ ions requires a significant amount of energy to overcome, leading to a high melting point.
-
Hardness and Brittleness: The rigid, ordered arrangement of ions in the crystal lattice contributes to MgO's hardness. However, the strong ionic bonds also make it brittle; any displacement of ion layers leads to strong repulsive forces, causing fractures.
-
Solubility: MgO's solubility varies depending on the solvent. It's relatively insoluble in water but dissolves in acidic solutions, where the oxide ions react with hydrogen ions (H⁺) to form water.
Applications of Magnesium Oxide (MgO)
Magnesium oxide's unique properties make it invaluable in a wide range of applications:
-
Refractory Materials: MgO's high melting point and chemical inertness make it ideal for use in high-temperature applications, such as furnace linings and crucibles.
-
Medicine: MgO is used as an antacid and laxative. Its ability to neutralize stomach acid is directly related to the basic nature of the oxide ion.
-
Agriculture: MgO serves as a source of magnesium, an essential nutrient for plant growth.
-
Electronics: MgO is employed in various electronic components, leveraging its electrical insulating properties.
-
Environmental applications: MgO has been investigated for its potential in carbon capture and storage.
Beyond the Basics: Exploring Advanced Concepts
While the electron dot structure provides a fundamental understanding of MgO, more advanced concepts provide a richer picture:
-
Formal Charges: Assigning formal charges to the ions in MgO helps further elucidate the stability of the ionic bond.
-
Lattice Energy: This thermodynamic quantity quantifies the energy released when gaseous ions combine to form a crystalline solid. For MgO, the high lattice energy reflects the strength of the ionic bonds.
-
Polarizability: While MgO is an ionic compound, some degree of polarizability exists due to the deformability of the electron clouds.
-
Band Theory: At a more advanced level, band theory can explain the electrical properties of MgO, revealing its insulating nature.
Conclusion: The Power of Simplicity and the Depth of Understanding
The electron dot structure for magnesium oxide, while a simplified representation, serves as a powerful entry point into understanding ionic bonding and the properties of this important compound. By grasping the fundamental principles illustrated by this structure, we gain a deeper appreciation for the intricate interplay of electrons, ions, and their collective behavior in shaping the macroscopic world. From its use in high-temperature applications to its role in biological systems, MgO's versatility highlights the significance of understanding its underlying chemical structure. This article has only scratched the surface of this fascinating compound, and further exploration will undoubtedly reveal even more insights into its properties and applications.
Latest Posts
Latest Posts
-
Which Valve Prevents Backflow Into The Left Ventricle
Apr 05, 2025
-
Cervical Vertebrae Differ From Other Vertebrae In What Way
Apr 05, 2025
-
What Is The Greatest Negative Integer
Apr 05, 2025
-
The Energy That Drives The Water Cycle Comes From The
Apr 05, 2025
-
Why Is Noise Pollution Regarded As Harmful For Human Beings
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Electron Dot Structure For Magnesium Oxide . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
