Determine The Quantity Of Molecules In 2.00 Moles Of P4
News Leon
Apr 02, 2025 · 5 min read
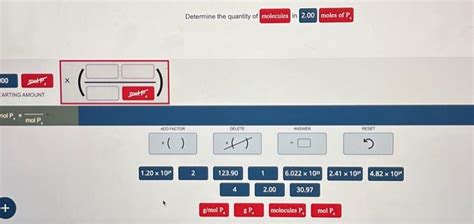
Table of Contents
Determining the Quantity of Molecules in 2.00 Moles of P₄: A Deep Dive into Mole Concepts
Understanding the relationship between moles, molecules, and Avogadro's number is fundamental in chemistry. This article will comprehensively explore how to determine the quantity of molecules in 2.00 moles of P₄, delving into the underlying concepts and providing a step-by-step guide. We'll also touch upon related calculations and the significance of Avogadro's number in stoichiometry.
Understanding Moles and Avogadro's Number
Before we delve into the calculation, let's establish a strong foundation in the core concepts.
What is a mole? A mole (mol) is a fundamental unit in chemistry that represents a specific number of particles, be it atoms, molecules, ions, or formula units. This number is known as Avogadro's number, approximately 6.022 x 10²³. One mole of any substance contains Avogadro's number of particles. Think of it like a dozen—a dozen eggs always means 12 eggs, regardless of the type of eggs. Similarly, one mole of any substance contains 6.022 x 10²³ particles.
Avogadro's Number: The Cornerstone of Chemistry
Amedeo Avogadro's contribution to chemistry is immeasurable. Avogadro's number is a constant that links the macroscopic world (grams, moles) to the microscopic world (atoms, molecules). It allows us to seamlessly convert between the mass of a substance and the number of particles it contains. This is crucial for performing stoichiometric calculations, which are essential for understanding chemical reactions and their quantitative aspects.
Molar Mass: Connecting Moles and Mass
The molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It's numerically equal to the atomic weight (for elements) or the molecular weight (for compounds) found on the periodic table. For example, the molar mass of carbon (C) is approximately 12.01 g/mol, while the molar mass of water (H₂O) is approximately 18.02 g/mol (1.01 x 2 + 16.00). Knowing the molar mass allows us to convert between the mass of a substance and the number of moles present.
Calculating Molecules in 2.00 Moles of P₄
Now, let's address the central question: how many molecules are present in 2.00 moles of P₄ (tetraphosphorus)?
Step 1: Understanding the Chemical Formula
The chemical formula P₄ indicates that each molecule of tetraphosphorus consists of four phosphorus atoms covalently bonded together. This is crucial for understanding the relationship between moles of P₄ and the number of molecules.
Step 2: Applying Avogadro's Number
Since one mole of any substance contains Avogadro's number of particles, 2.00 moles of P₄ will contain twice that many molecules:
2.00 moles P₄ × (6.022 x 10²³ molecules P₄ / 1 mole P₄) = 1.2044 x 10²⁴ molecules P₄
Therefore, there are approximately 1.2044 x 10²⁴ molecules in 2.00 moles of P₄.
Extending the Concept: Calculations Involving Mass and Molar Mass
Let's expand our understanding by incorporating the concept of molar mass. Suppose we want to determine the number of molecules in a given mass of P₄.
Example: How many molecules are present in 124 g of P₄?
Step 1: Calculate the Molar Mass of P₄
The atomic mass of phosphorus (P) is approximately 30.97 g/mol. Therefore, the molar mass of P₄ is:
4 x 30.97 g/mol = 123.88 g/mol
Step 2: Convert Grams to Moles
We can use the molar mass to convert the given mass (124 g) to moles:
124 g P₄ × (1 mole P₄ / 123.88 g P₄) ≈ 1.00 moles P₄
Step 3: Calculate the Number of Molecules
Now, we can use Avogadro's number to determine the number of molecules:
1.00 moles P₄ × (6.022 x 10²³ molecules P₄ / 1 mole P₄) ≈ 6.022 x 10²³ molecules P₄
Therefore, there are approximately 6.022 x 10²³ molecules in 124 g of P₄.
Beyond P₄: Applying the Concepts to Other Substances
The principles discussed here are applicable to any substance, whether it's an element, a compound, or an ion. The only difference lies in the molar mass and the interpretation of the "particle". For example:
- For elemental substances: The particle is an atom.
- For molecular compounds: The particle is a molecule.
- For ionic compounds: The particle is a formula unit.
The Importance of Significant Figures
Throughout these calculations, it's crucial to pay attention to significant figures. The number of significant figures in the final answer should reflect the least precise measurement used in the calculation. In our example using 2.00 moles, we maintained three significant figures throughout.
Applications in Stoichiometry and Chemical Reactions
Understanding mole concepts and Avogadro's number is pivotal for stoichiometry, which deals with the quantitative relationships between reactants and products in chemical reactions. Being able to calculate the number of molecules involved allows chemists to accurately predict the amounts of substances needed or produced in a reaction. This is crucial in various applications, from industrial chemical processes to laboratory experiments.
Conclusion: Mastering Mole Calculations
The ability to determine the quantity of molecules in a given amount of substance is a fundamental skill in chemistry. By understanding the concepts of moles, Avogadro's number, and molar mass, and by applying the appropriate conversion factors, we can confidently perform these calculations. This article has provided a comprehensive guide, including examples and explanations, to solidify your understanding of this important aspect of chemistry. Remember to always pay close attention to significant figures and apply the principles consistently to diverse chemical scenarios. The ability to accurately perform these calculations is critical for success in many chemistry-related fields and endeavors.
Latest Posts
Latest Posts
-
Is Delta H Positive For Endothermic
Apr 03, 2025
-
Cytoplasm Of The Cell Is Being Divided
Apr 03, 2025
-
Which Elements Has Only One Valence Electron
Apr 03, 2025
-
Co2 Enters The Inner Spaces Of The Leaf Through The
Apr 03, 2025
-
Which Of The Following Is An Alcohol
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Determine The Quantity Of Molecules In 2.00 Moles Of P4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
