Which Of The Following Is An Alcohol
News Leon
Apr 03, 2025 · 6 min read
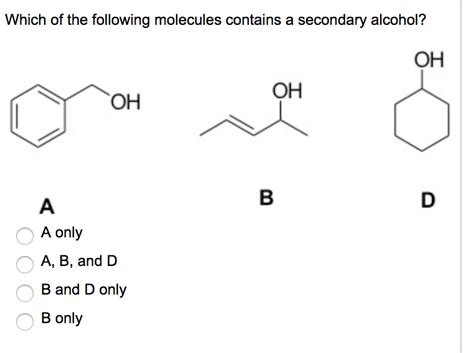
Table of Contents
Which of the Following is an Alcohol? Understanding the Chemistry and Properties of Alcohols
The question, "Which of the following is an alcohol?" might seem simple at first glance. However, a deep dive into the chemistry of alcohols reveals a fascinating world of organic compounds with diverse properties and applications. This article will not only answer this question in various contexts, but will also provide a comprehensive understanding of alcohols, their classification, properties, and significance.
What Defines an Alcohol?
Before we can identify which compound is an alcohol, we need a clear definition. In organic chemistry, an alcohol is an organic compound containing a hydroxyl group (-OH) bonded to a saturated carbon atom. This seemingly simple definition holds the key to understanding the vast array of molecules classified as alcohols. The hydroxyl group is the functional group responsible for many of the characteristic properties of alcohols.
The saturated carbon atom means that the carbon atom bonded to the hydroxyl group is only bonded to single bonds (no double or triple bonds). This distinction separates alcohols from other oxygen-containing compounds like phenols (where the hydroxyl group is attached to a benzene ring) and enols (where the hydroxyl group is attached to a carbon atom that's also double bonded to another carbon atom).
Identifying Alcohols: A Step-by-Step Approach
To correctly identify an alcohol from a list of compounds, follow these steps:
-
Identify the Hydroxyl Group: Look for the -OH group within the molecular structure.
-
Check the Carbon Atom: Verify that the carbon atom bonded to the -OH group is saturated (only single bonds).
-
Consider Exceptions: Be aware of exceptions and related compounds like phenols and enols, which, while containing hydroxyl groups, aren't classified as alcohols.
Let's look at some examples:
-
Ethanol (CH3CH2OH): This is a classic example of an alcohol. The hydroxyl group (-OH) is attached to a saturated carbon atom (the ethyl group).
-
Methanol (CH3OH): Another common alcohol, with the hydroxyl group attached to a methyl group (a saturated carbon).
-
Isopropanol (CH3CH(OH)CH3): This is an example of a secondary alcohol, where the carbon atom bearing the hydroxyl group is attached to two other carbon atoms.
-
Phenol (C6H5OH): Although containing a hydroxyl group, phenol is not an alcohol. The hydroxyl group is attached to an aromatic ring (benzene), making it a phenol.
-
Ethylene Glycol (HOCH2CH2OH): This is a diol, meaning it contains two hydroxyl groups, one on each carbon atom. It's still classified as an alcohol.
-
Glycerol (HOCH2CH(OH)CH2OH): This is a triol containing three hydroxyl groups. Again, it remains within the alcohol classification.
Classification of Alcohols
Alcohols are classified based on the number of carbon atoms attached to the carbon atom bearing the hydroxyl group:
-
Primary Alcohols: The carbon atom bonded to the -OH group is only attached to one other carbon atom. Examples include methanol and ethanol. These are typically more reactive in oxidation reactions.
-
Secondary Alcohols: The carbon atom bonded to the -OH group is attached to two other carbon atoms. Isopropanol is a good example. These alcohols can also be oxidized, but to form ketones.
-
Tertiary Alcohols: The carbon atom bonded to the -OH group is attached to three other carbon atoms. Tertiary alcohols are resistant to oxidation.
Properties of Alcohols
The properties of alcohols are largely determined by the presence of the hydroxyl group and the size and structure of the alkyl group (the carbon chain attached to the -OH).
Physical Properties:
-
Polarity: The hydroxyl group is highly polar due to the electronegativity difference between oxygen and hydrogen, resulting in hydrogen bonding. This leads to higher boiling points compared to hydrocarbons of similar molecular weight.
-
Solubility: Lower molecular weight alcohols (like methanol and ethanol) are miscible with water due to the strong hydrogen bonding between the alcohol and water molecules. As the size of the alkyl group increases, solubility in water decreases.
-
Density: Generally, alcohols have higher densities than hydrocarbons but lower densities than water.
Chemical Properties:
-
Acidity: Alcohols are weakly acidic, meaning they can donate a proton (H+). The acidity increases with the increasing number of electron-withdrawing groups attached to the carbon atom bearing the -OH group.
-
Basicity: Alcohols can also act as weak bases, accepting a proton. This is due to the lone pair of electrons on the oxygen atom.
-
Oxidation: Primary alcohols can be oxidized to aldehydes and then to carboxylic acids. Secondary alcohols are oxidized to ketones. Tertiary alcohols are resistant to oxidation.
-
Esterification: Alcohols react with carboxylic acids to form esters, an important class of organic compounds with various applications, including fragrances and flavorings.
-
Dehydration: Alcohols can undergo dehydration reactions, losing a water molecule to form alkenes. The conditions required for dehydration depend on the type of alcohol (primary, secondary, or tertiary).
Applications of Alcohols
Alcohols have a wide range of applications across various industries:
-
Solvents: Alcohols are excellent solvents for many organic compounds and are used in paints, varnishes, and inks. Ethanol is commonly used as a solvent in pharmaceuticals and cosmetics.
-
Fuels: Ethanol is a biofuel, blended with gasoline to reduce dependence on fossil fuels. Methanol is used as a fuel in some applications.
-
Pharmaceuticals: Many pharmaceuticals are alcohols or contain alcohol groups as part of their structure. Ethanol is used as an antiseptic and disinfectant. Isopropanol is a common ingredient in hand sanitizers.
-
Cosmetics and Personal Care Products: Alcohols are used as solvents and preservatives in cosmetics and personal care products.
-
Food and Beverages: Ethanol is the active ingredient in alcoholic beverages. Other alcohols are used as flavoring agents and preservatives.
-
Industrial Processes: Alcohols are used as intermediates in the synthesis of various chemicals, including plastics, resins, and fibers.
Identifying Alcohols in a Given Set: Examples
Let's consider a few examples of identifying alcohols within a list of compounds:
Example 1:
Which of the following is an alcohol?
a) CH3CH2Cl (Chloroethane) b) CH3CH2OH (Ethanol) c) CH3COOH (Acetic acid) d) CH3CHO (Acetaldehyde)
Answer: b) CH3CH2OH (Ethanol) is the alcohol. It has the -OH group attached to a saturated carbon.
Example 2:
Which of the following is NOT an alcohol?
a) CH3CH2CH2OH (Propanol) b) (CH3)3COH (tert-Butanol) c) C6H5OH (Phenol) d) CH3CH(OH)CH3 (Isopropanol)
Answer: c) C6H5OH (Phenol) is not an alcohol. The hydroxyl group is attached to an aromatic ring.
Example 3:
Which of the following is a secondary alcohol?
a) CH3OH (Methanol) b) CH3CH2CH2OH (Propanol) c) CH3CH(OH)CH3 (Isopropanol) d) (CH3)3COH (tert-Butanol)
Answer: c) CH3CH(OH)CH3 (Isopropanol) is a secondary alcohol; the carbon atom with the -OH group is attached to two other carbons.
Conclusion
Understanding what constitutes an alcohol goes beyond simply recognizing the -OH group. The nature of the carbon atom to which it is bonded is crucial. This article provides a comprehensive overview of alcohols, their properties, classification, and extensive applications. By following the steps outlined for identification and understanding the chemical properties, one can confidently determine which compound amongst a set belongs to this vital class of organic compounds. The ability to identify and classify alcohols is essential for anyone studying organic chemistry or working in fields where these compounds are used. The vast applications of alcohols highlight their importance in various industries, showcasing their multifaceted role in modern life.
Latest Posts
Latest Posts
-
Paleolithic Age Is Also Known As The
Apr 04, 2025
-
What Is Found In Both Eukaryotic And Prokaryotic Cells
Apr 04, 2025
-
Select The Correct Electron Configuration For Cr
Apr 04, 2025
-
Can A Pure Substance Be A Compound
Apr 04, 2025
-
3 Types Of Winds Class 7
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is An Alcohol . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
