Composed Of Long Chains Of Amino Acids
News Leon
Apr 04, 2025 · 6 min read
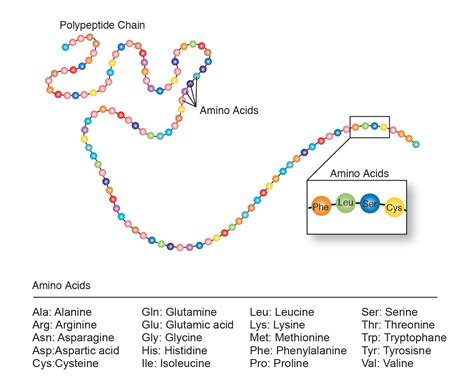
Table of Contents
Composed of Long Chains of Amino Acids: Exploring Proteins and Their Biological Significance
Proteins: the workhorses of the cell. This seemingly simple phrase belies the incredible complexity and diversity of these macromolecules, the building blocks of life itself. Composed of long chains of amino acids, proteins perform a vast array of functions essential for life, from catalyzing biochemical reactions to providing structural support and mediating cell signaling. Understanding their structure and function is crucial to comprehending the intricacies of biology, medicine, and biotechnology.
The Building Blocks: Amino Acids
The fundamental units of proteins are amino acids. These organic molecules are characterized by a central carbon atom (the α-carbon) bonded to four distinct groups:
- An amino group (-NH₂): This group is basic and contributes to the protein's overall charge.
- A carboxyl group (-COOH): This group is acidic and also influences the protein's charge.
- A hydrogen atom (-H): A simple hydrogen atom.
- A side chain (R-group): This is the variable group, differing among the 20 standard amino acids, and dictates each amino acid's unique properties.
These R-groups are the key to the incredible diversity of proteins. They can be:
- Nonpolar (hydrophobic): These tend to cluster together in the interior of a protein, away from water. Examples include alanine, valine, and leucine.
- Polar (hydrophilic): These interact readily with water and are often found on the protein's surface. Examples include serine, threonine, and asparagine.
- Charged (ionic): These possess either a positive or negative charge at physiological pH, influencing protein-protein interactions and enzyme activity. Examples include lysine (positive) and aspartic acid (negative).
- Aromatic: These contain ring structures and often absorb ultraviolet light. Examples include phenylalanine, tyrosine, and tryptophan.
The unique properties of each R-group influence the overall three-dimensional structure and function of the protein.
Peptide Bonds: Linking Amino Acids
Amino acids are linked together via peptide bonds, a type of covalent bond formed between the carboxyl group of one amino acid and the amino group of another. This reaction releases a molecule of water (a dehydration reaction). A chain of amino acids linked by peptide bonds is called a polypeptide. Proteins can consist of one or more polypeptide chains.
Protein Structure: A Hierarchy of Organization
The structure of a protein is intimately linked to its function. Protein structure is often described using a four-level hierarchy:
1. Primary Structure: The Amino Acid Sequence
The primary structure of a protein refers to the linear sequence of amino acids in the polypeptide chain. This sequence is dictated by the genetic code, and even a single amino acid substitution can dramatically alter the protein's structure and function (as famously seen in sickle cell anemia). The primary structure is the foundation upon which all higher levels of structure are built.
2. Secondary Structure: Local Folding Patterns
The secondary structure refers to local folding patterns within the polypeptide chain. These patterns are stabilized by hydrogen bonds between the backbone atoms (not the R-groups). Common secondary structures include:
- α-helices: A right-handed coiled structure stabilized by hydrogen bonds between the carbonyl oxygen of one amino acid and the amide hydrogen of another amino acid four residues down the chain.
- β-sheets: Extended, sheet-like structures formed by hydrogen bonding between polypeptide strands that can be parallel or antiparallel.
- Random coils or loops: Regions of the polypeptide chain that lack a defined secondary structure.
3. Tertiary Structure: The Three-Dimensional Arrangement
The tertiary structure describes the overall three-dimensional arrangement of a polypeptide chain. This structure is determined by various interactions between the R-groups of the amino acids, including:
- Hydrophobic interactions: Nonpolar R-groups cluster together in the protein's interior, minimizing their contact with water.
- Hydrogen bonds: Interactions between polar R-groups.
- Ionic bonds (salt bridges): Electrostatic interactions between oppositely charged R-groups.
- Disulfide bonds: Covalent bonds between cysteine residues, creating strong cross-links within the protein.
The tertiary structure dictates the protein's overall shape and function. A change in even one amino acid can disrupt the tertiary structure and lead to protein misfolding.
4. Quaternary Structure: Multiple Polypeptide Chains
Some proteins are composed of multiple polypeptide chains, each with its own tertiary structure. The arrangement of these subunits forms the quaternary structure. Examples include hemoglobin (four subunits) and many enzymes. Interactions similar to those in tertiary structure stabilize the quaternary structure.
Protein Function: A Diverse Repertoire
Proteins are incredibly versatile molecules, performing a vast array of functions essential for life. Some key roles include:
1. Enzymes: Catalyzing Biochemical Reactions
Enzymes are biological catalysts that accelerate the rate of biochemical reactions. They do this by lowering the activation energy required for the reaction to proceed. Their specificity is remarkable; each enzyme typically catalyzes only one type of reaction. The active site of an enzyme, a specific region on its surface, binds the substrate (the molecule being acted upon) and facilitates the reaction.
2. Structural Proteins: Providing Support and Shape
Structural proteins provide support and shape to cells and tissues. Examples include collagen (found in connective tissue), keratin (found in hair and nails), and tubulin (a component of microtubules). These proteins often have fibrous structures, providing strength and resilience.
3. Transport Proteins: Moving Molecules Across Membranes
Transport proteins facilitate the movement of molecules across cell membranes. Membrane proteins, such as channels and carriers, selectively transport specific molecules across the lipid bilayer. This is essential for maintaining cellular homeostasis. Examples include ion channels and glucose transporters.
4. Motor Proteins: Generating Movement
Motor proteins are responsible for generating movement within cells and tissues. Examples include myosin (involved in muscle contraction) and kinesin (involved in intracellular transport). These proteins often undergo conformational changes to produce movement.
5. Signaling Proteins: Mediating Cell Communication
Signaling proteins mediate cell communication by transmitting signals between cells or within a cell. Hormones, neurotransmitters, and receptors are examples of signaling proteins. These proteins often interact with other molecules to trigger downstream responses.
6. Defense Proteins: Protecting Against Pathogens
Defense proteins protect the body from pathogens (disease-causing organisms). Antibodies, components of the immune system, are examples of defense proteins that recognize and neutralize foreign invaders.
Protein Misfolding and Disease
The proper folding of proteins is crucial for their function. Errors in protein folding can lead to the formation of misfolded proteins, which can aggregate and form amyloid fibrils. These aggregates are associated with numerous diseases, including:
- Alzheimer's disease: Characterized by the accumulation of amyloid-beta plaques in the brain.
- Parkinson's disease: Associated with the aggregation of alpha-synuclein protein in the brain.
- Type II diabetes: Linked to the aggregation of amylin in the pancreas.
- Huntington's disease: Caused by the aggregation of huntingtin protein.
Understanding the mechanisms of protein misfolding and aggregation is crucial for the development of therapies for these devastating diseases.
Conclusion: The Ever-Evolving World of Proteins
Proteins, composed of long chains of amino acids, are the versatile workhorses of the cell, carrying out a myriad of functions essential for life. Their intricate structures, determined by the amino acid sequence and various interactions, underpin their diverse roles in enzymatic catalysis, structural support, transport, movement, signaling, and defense. The study of proteins remains a vibrant field, constantly revealing new insights into their complexity and biological significance, with profound implications for medicine, biotechnology, and our understanding of life itself. Future research will undoubtedly uncover even more about the intricacies of protein structure, function, and their roles in health and disease, paving the way for innovative therapeutic interventions and biotechnological applications. The journey into the world of proteins is far from over; the more we learn, the more we realize the depth and breadth of their remarkable impact on the living world.
Latest Posts
Latest Posts
-
20 Of 2 Is Equal To
Apr 05, 2025
-
Which Of The Following Is Not An Internet Protocol
Apr 05, 2025
-
Sum Of The Exterior Angles Of A Decagon
Apr 05, 2025
-
What Are Characteristics Of Covalent Compounds
Apr 05, 2025
-
What Is The Name Of The Following
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Composed Of Long Chains Of Amino Acids . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
