What Are Characteristics Of Covalent Compounds
News Leon
Apr 05, 2025 · 6 min read
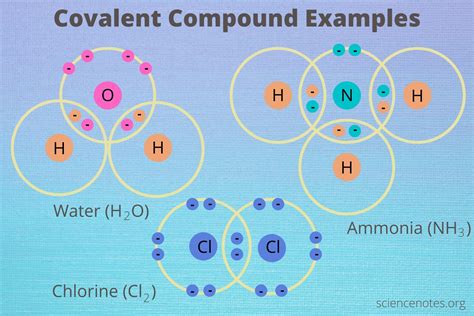
Table of Contents
Characteristics of Covalent Compounds: A Comprehensive Guide
Covalent compounds, also known as molecular compounds, represent a significant class of chemical substances formed through the sharing of electrons between atoms. Understanding their unique characteristics is crucial in various fields, from chemistry and biology to materials science and engineering. This comprehensive guide delves into the defining features of covalent compounds, exploring their properties, bonding mechanisms, and applications.
Defining Covalent Bonding: The Essence of Sharing
At the heart of covalent compounds lies the covalent bond, a powerful chemical link forged when two or more atoms share one or more pairs of valence electrons. Unlike ionic bonds, where electrons are transferred from one atom to another, covalent bonds involve a mutual sharing of electrons to achieve a stable electron configuration, typically resembling that of a noble gas. This sharing creates a strong attractive force that holds the atoms together, forming molecules.
Types of Covalent Bonds: A Spectrum of Sharing
Covalent bonds aren't monolithic; they exhibit a spectrum of characteristics based on the nature of electron sharing:
-
Nonpolar Covalent Bonds: In these bonds, the electrons are shared almost equally between atoms of similar electronegativity. Electronegativity refers to an atom's ability to attract electrons towards itself. Examples include bonds within diatomic molecules like O₂ (oxygen) and N₂ (nitrogen).
-
Polar Covalent Bonds: Here, the electrons are shared unequally between atoms with differing electronegativities. This unequal sharing creates a dipole moment, a separation of positive and negative charges within the molecule. Water (H₂O) is a classic example, with oxygen attracting electrons more strongly than hydrogen.
-
Coordinate Covalent Bonds (Dative Bonds): These bonds involve one atom donating both electrons to the shared pair. This is often seen in complex ions and molecules containing transition metals. An example can be found in the ammonium ion (NH₄⁺).
Physical Properties of Covalent Compounds: A Diverse Landscape
Covalent compounds display a remarkable range of physical properties, largely dictated by the nature of their bonding and intermolecular forces:
1. Low Melting and Boiling Points:
Generally, covalent compounds have relatively low melting and boiling points compared to ionic compounds. This is because the intermolecular forces (forces between molecules) in covalent compounds are weaker than the strong electrostatic forces present in ionic lattices. Less energy is required to overcome these weaker forces, resulting in lower melting and boiling points. Consider the contrast between table salt (NaCl, high melting point) and sugar (sucrose, a covalent compound with a lower melting point).
2. Poor Electrical Conductivity:
Most covalent compounds are poor conductors of electricity in both solid and liquid states. This is because they lack freely mobile charged particles (ions or electrons) to carry electrical current. In contrast, molten ionic compounds and their aqueous solutions are good conductors due to the presence of ions.
3. Variable Solubility:
The solubility of covalent compounds varies widely depending on the polarity of the molecule and the solvent used. Polar covalent compounds tend to dissolve well in polar solvents (like water), while nonpolar covalent compounds dissolve better in nonpolar solvents (like oil). This is based on the principle of "like dissolves like."
4. Lower Hardness and Brittleness:
Covalent compounds are generally softer and more brittle than ionic compounds. This is because the relatively weak intermolecular forces in covalent compounds make them less resistant to deformation and fracture.
5. Different States of Matter:
Covalent compounds can exist in all three common states of matter – solid, liquid, and gas – at room temperature depending on the strength of their intermolecular forces and molecular weight. For example, water is a liquid, carbon dioxide is a gas, and sugar is a solid.
Chemical Properties of Covalent Compounds: Reactivity and Transformations
The chemical behavior of covalent compounds is dictated by several factors, including bond strength, polarity, and the presence of functional groups.
1. Combustion:
Many covalent compounds, particularly those containing carbon and hydrogen (hydrocarbons), readily undergo combustion reactions in the presence of oxygen, producing carbon dioxide and water. This reaction is highly exothermic, releasing significant amounts of energy.
2. Reactions with Acids and Bases:
Depending on their structure and functional groups, covalent compounds can react with acids and bases. For instance, carboxylic acids can react with bases to form salts, while esters can undergo hydrolysis reactions in the presence of acids or bases.
3. Substitution and Addition Reactions:
Organic covalent compounds (carbon-based) frequently participate in substitution reactions (one atom or group replacing another) and addition reactions (atoms or groups adding to a molecule), leading to the formation of new covalent bonds.
4. Polymerization:
Many covalent compounds can undergo polymerization, a process where small molecules (monomers) link together to form large chain-like molecules (polymers). This process is fundamental to the creation of plastics, fibers, and other synthetic materials.
Examples of Covalent Compounds and Their Applications: A World of Possibilities
Covalent compounds are ubiquitous, playing vital roles in diverse applications:
1. Organic Chemistry and Biochemistry:
The vast majority of organic molecules – including carbohydrates, lipids, proteins, and nucleic acids – are covalent compounds. These molecules form the basis of life itself, performing critical functions in metabolism, structure, and information storage.
2. Polymers and Plastics:
Polymers, including polyethylene (plastic bags), polypropylene (containers), and polystyrene (foam cups), are composed of long chains of covalently bonded atoms. These materials are essential in numerous industries, from packaging to construction.
3. Pharmaceuticals:
Many pharmaceuticals are covalent compounds meticulously designed to interact with specific biological targets, such as enzymes or receptors. These drugs exert their therapeutic effects by forming covalent or non-covalent interactions with their targets.
4. Semiconductors:
Silicon and other semiconducting materials are based on covalent bonding arrangements, enabling their unique electrical properties crucial for electronic devices.
5. Fuels:
Hydrocarbons, covalent compounds consisting primarily of carbon and hydrogen atoms, serve as primary fuels for transportation, heating, and electricity generation.
Distinguishing Covalent from Ionic Compounds: A Key Differentiation
It's important to distinguish covalent compounds from ionic compounds, which are characterized by the electrostatic attraction between oppositely charged ions. Here's a summary of key differences:
| Feature | Covalent Compounds | Ionic Compounds |
|---|---|---|
| Bonding | Sharing of electrons | Transfer of electrons |
| Melting Point | Generally low | Generally high |
| Boiling Point | Generally low | Generally high |
| Electrical Conductivity | Poor (except for graphite) | Good when molten or dissolved in water |
| Solubility | Varies, depends on polarity | Generally soluble in polar solvents |
| Hardness | Generally soft and brittle | Generally hard and brittle |
| Crystalline Structure | Molecular structures | Ionic lattices |
Conclusion: The Enduring Importance of Covalent Compounds
Covalent compounds constitute a cornerstone of chemistry, encompassing a vast array of molecules with diverse structures and properties. Their importance extends far beyond the laboratory, impacting numerous aspects of our daily lives, from the food we eat to the technologies we use. By understanding their fundamental characteristics, we can harness their potential for innovation and advancement in various scientific and technological domains. Further exploration into the intricacies of covalent bonding and their myriad applications will undoubtedly unlock new possibilities in the future.
Latest Posts
Latest Posts
-
What Three Elements Make Up Carbohydrates
Apr 05, 2025
-
The Movement Of People From One Place To Another
Apr 05, 2025
-
Sample Of An Authorization Letter To Claim A Document
Apr 05, 2025
-
All Of The Following Are Eukaryotic Except
Apr 05, 2025
-
A Place In The Body Where Two Bones Come Together
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Are Characteristics Of Covalent Compounds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
