Cobalt Ii Hydrogen Carbonate Chemical Formula
News Leon
Apr 02, 2025 · 6 min read
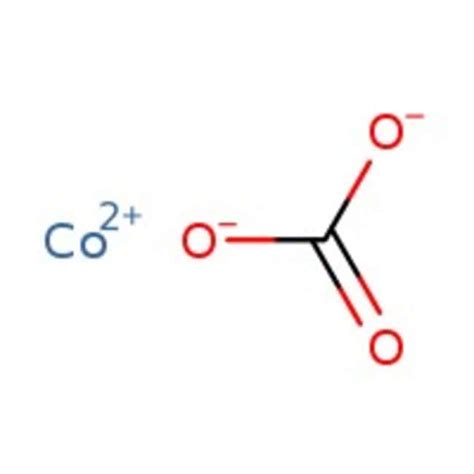
Table of Contents
Cobalt(II) Hydrogen Carbonate: A Deep Dive into its Chemical Formula, Properties, and Applications
Cobalt(II) hydrogen carbonate, while not as commonly encountered as some other cobalt compounds, holds significant importance in various chemical and industrial processes. Understanding its chemical formula, properties, and applications is crucial for anyone working with this compound or related fields. This comprehensive article delves deep into the intricacies of cobalt(II) hydrogen carbonate, exploring its characteristics and uses in detail.
Understanding the Chemical Formula: Co(HCO₃)₂
The chemical formula for cobalt(II) hydrogen carbonate is Co(HCO₃)₂. This formula reveals key aspects of the compound's composition:
-
Co: This represents the cobalt(II) ion, indicating that cobalt is present in its +2 oxidation state. Cobalt, a transition metal, exhibits variable oxidation states, but in this compound, it's in its +2 state.
-
HCO₃: This is the hydrogen carbonate ion (also known as bicarbonate), a polyatomic anion with a -1 charge. It's a crucial component in many chemical systems, including natural water systems and biological processes.
The formula Co(HCO₃)₂ signifies that one cobalt(II) ion is bound to two hydrogen carbonate ions to maintain electrical neutrality. The overall charge of the compound is zero.
Importance of Oxidation State
The Roman numeral II in "Cobalt(II)" is crucial. It specifies the oxidation state of cobalt, differentiating it from other possible cobalt oxidation states like +3 (Cobalt(III)). The oxidation state heavily influences the compound's properties, reactivity, and applications. A misunderstanding of the oxidation state could lead to incorrect predictions about its behavior.
Physical and Chemical Properties of Cobalt(II) Hydrogen Carbonate
Cobalt(II) hydrogen carbonate, like many transition metal carbonates, exhibits specific physical and chemical properties:
Physical Properties:
-
Appearance: While a pure sample isn't readily available for visual description due to its instability, it's likely a pink or reddish solid, reflecting the typical color of cobalt(II) compounds. The exact appearance could vary depending on hydration levels (presence of water molecules).
-
Solubility: It's expected to have limited solubility in water, similar to other metal hydrogen carbonates. The solubility might be influenced by pH and temperature.
-
Stability: Cobalt(II) hydrogen carbonate is known to be unstable. It readily decomposes, especially in the presence of heat or changes in pH. This decomposition often leads to the formation of cobalt(II) carbonate, carbon dioxide, and water. This instability is a significant factor to consider when handling or using this compound.
Chemical Properties:
-
Decomposition: As mentioned, its primary chemical characteristic is its tendency to decompose. This reaction can be represented as follows:
Co(HCO₃)₂ → CoCO₃ + H₂O + CO₂
-
Reactions with Acids: Cobalt(II) hydrogen carbonate reacts with acids, producing cobalt(II) salts, water, and carbon dioxide. The specific reaction depends on the acid used. For instance, with hydrochloric acid (HCl), the reaction would be:
Co(HCO₃)₂ + 2HCl → CoCl₂ + 2H₂O + 2CO₂
-
Reactions with Bases: Reactions with bases lead to the formation of cobalt(II) hydroxide and carbonate salts. The exact products depend on the base's strength and concentration.
-
Oxidation States: The +2 oxidation state of cobalt is relatively stable in this compound, but under certain conditions, oxidation to a higher oxidation state (+3) is possible, though unlikely under normal circumstances.
Synthesis of Cobalt(II) Hydrogen Carbonate
Preparing pure cobalt(II) hydrogen carbonate directly is challenging due to its instability. The common approach involves indirect methods or obtaining it as an intermediate product during other reactions. One possible route might involve reacting a cobalt(II) salt (like cobalt(II) chloride or sulfate) with a solution of sodium hydrogen carbonate (NaHCO₃) under carefully controlled conditions of temperature and pH. However, isolating the pure compound would be problematic due to its immediate decomposition.
The difficulty in synthesizing and isolating pure Co(HCO₃)₂ highlights the importance of understanding its inherent instability and the likely presence of decomposition products in any preparation attempts.
Applications of Cobalt(II) Hydrogen Carbonate
Despite its instability, cobalt(II) hydrogen carbonate finds limited, albeit crucial, applications in niche areas:
-
Precursor for other Cobalt Compounds: Due to its instability, it's more likely to serve as a transient intermediate in the synthesis of other more stable cobalt compounds. This could be an important aspect in specific chemical synthesis pathways where the hydrogen carbonate might act as a temporary carrier of cobalt.
-
Catalysis (Potential Application): The potential application in catalysis might exist in specific reactions. Its decomposition products, particularly cobalt(II) carbonate and cobalt(II) oxide, are well-known catalysts in various industrial processes. Therefore, while Co(HCO₃)₂ itself might not be the catalyst, it could be a source material for generating catalytic species in situ.
-
Electrochemical Applications (Potential Application): The possibility of exploring its use in electrochemical processes related to cobalt-based batteries and other electrochemical devices exists. However, its instability and the need for controlled conditions will be major obstacles.
-
Research Applications: Its limited application is largely restricted to research settings where its properties and reactions are studied in controlled conditions. This helps to improve our fundamental understanding of cobalt chemistry and reaction mechanisms.
Safety Precautions When Handling Cobalt Compounds
Cobalt compounds, including cobalt(II) hydrogen carbonate, should be handled with care. The following safety precautions are essential:
-
Avoid Inhalation: Cobalt compounds can be harmful if inhaled, so proper ventilation and respiratory protection are essential.
-
Skin and Eye Contact: Avoid contact with skin and eyes. Wear appropriate personal protective equipment (PPE) such as gloves, goggles, and lab coats.
-
Ingestion: Never ingest cobalt compounds.
-
Waste Disposal: Dispose of cobalt-containing waste according to local regulations and guidelines.
Future Research Directions
Further research on cobalt(II) hydrogen carbonate could explore the following aspects:
-
Improved Synthesis Methods: Developing more efficient and controlled methods for the synthesis and isolation of this unstable compound.
-
Detailed Characterization: Employing advanced techniques to fully characterize its properties, including structural analysis, thermal analysis, and spectroscopic studies.
-
Exploration of Catalytic Applications: Investigating its potential application as a precursor or catalyst in various chemical reactions.
-
Electrochemical Behavior: Understanding its electrochemical behavior and potential applications in energy storage or other electrochemical devices.
Conclusion
Cobalt(II) hydrogen carbonate, despite its inherent instability and limited direct applications, remains an important compound for understanding the chemistry of cobalt and its potential roles in various chemical processes. Although not directly used in many industrial settings, its role as a precursor and potential catalyst warrants further investigation. Careful handling and adherence to safety protocols are crucial when working with this compound or any cobalt-containing substance. Continued research in this area promises further insights into this fascinating, yet elusive, chemical species. The exploration of its properties and potential applications remains an active area of study with implications for catalysis, electrochemistry, and other fields of chemistry and materials science. Understanding its chemical formula, properties, and limitations is crucial for advancing knowledge and finding potential applications in different technologies.
Latest Posts
Latest Posts
-
Which Elements Has Only One Valence Electron
Apr 03, 2025
-
Co2 Enters The Inner Spaces Of The Leaf Through The
Apr 03, 2025
-
Which Of The Following Is An Alcohol
Apr 03, 2025
-
What Is The Smallest Odd Composite Number
Apr 03, 2025
-
Which Of The Following Represents A Deferral
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Cobalt Ii Hydrogen Carbonate Chemical Formula . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
