Cis 1 Tert Butyl 2 Methylcyclohexane
News Leon
Apr 02, 2025 · 6 min read
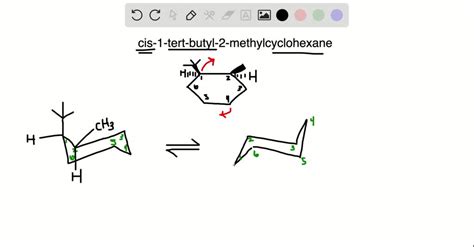
Table of Contents
Cis-1-tert-Butyl-2-methylcyclohexane: A Deep Dive into its Structure, Properties, and Significance
Cis-1-tert-butyl-2-methylcyclohexane is a fascinating organic compound that exemplifies the complexities of stereochemistry and conformational analysis. While seemingly simple in its chemical formula, understanding its unique structural features and resulting properties requires a detailed examination. This article delves into the intricacies of this molecule, exploring its stereochemistry, conformational preferences, spectroscopic characteristics, and potential applications.
Understanding the Molecular Structure
Cis-1-tert-butyl-2-methylcyclohexane's name itself reveals crucial structural information. Let's break it down:
-
Cyclohexane: This indicates a six-membered carbon ring forming a cycloalkane. Cyclohexane itself exists in various conformations, with the chair conformation being the most stable due to minimal steric strain.
-
1-tert-Butyl: This denotes a tert-butyl group (-(CH₃)₃C) attached to carbon atom number 1 in the cyclohexane ring. The tert-butyl group is significantly bulky.
-
2-Methyl: A methyl group (-CH₃) is attached to carbon atom number 2.
-
Cis: This is the critical stereochemical descriptor. It signifies that both the tert-butyl and methyl groups are on the same side of the cyclohexane ring. This contrasts with the trans isomer, where they would be on opposite sides.
The cis configuration significantly impacts the molecule's overall shape and properties, introducing steric interactions and influencing its conformational preferences.
Visualizing the Molecule
Imagine a cyclohexane ring in its chair conformation. In the cis isomer, both the bulky tert-butyl group and the smaller methyl group are pointing upwards (or both downwards) from the ring. This arrangement leads to significant steric hindrance.
Conformational Analysis: A Key to Understanding Properties
The chair conformation of cyclohexane is not static; it undergoes rapid interconversion between two equivalent chair forms through a process called ring flipping. However, the presence of the bulky tert-butyl and methyl groups significantly alters this equilibrium.
Steric Effects and Conformational Preference
The bulky tert-butyl group strongly prefers the equatorial position to minimize steric interactions with the axial hydrogens. Forcing it into the axial position leads to significant 1,3-diaxial interactions, making that conformation highly unfavorable.
In the cis-1-tert-butyl-2-methylcyclohexane, when the tert-butyl group occupies the equatorial position, the methyl group is necessarily forced into the axial position. While not as dramatically unfavorable as an axial tert-butyl group, this axial methyl group still contributes to steric strain.
Consequently, the most stable conformation of cis-1-tert-butyl-2-methylcyclohexane features the tert-butyl group equatorial and the methyl group axial. The alternative conformation (tert-butyl axial, methyl equatorial) is considerably less stable. The energy difference between these conformations is substantial, leading to a strong preference for the former.
Implications of Conformational Preference
This strong conformational preference has profound consequences for the molecule's physical and chemical properties:
-
Melting Point and Boiling Point: The steric interactions influence intermolecular forces, affecting the melting and boiling points. The predominantly equatorial tert-butyl group leads to less efficient packing in the solid state, likely resulting in a lower melting point compared to the trans isomer. Similarly, boiling point might be affected due to variations in intermolecular interactions.
-
Reactivity: The accessibility of the functional groups (tert-butyl and methyl) is directly influenced by their positions. The axial methyl group in the preferred conformation might exhibit slightly enhanced reactivity compared to an equatorial methyl group in certain reactions.
-
NMR Spectroscopy: Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful tool to probe molecular structure and conformation. The chemical shifts and coupling constants in the ¹H NMR spectrum will reflect the preferred conformation, providing experimental evidence supporting the computational predictions. The axial and equatorial protons will exhibit different chemical shifts due to their different magnetic environments.
Spectroscopic Characterization
Various spectroscopic techniques are crucial in confirming the structure and conformational preferences of cis-1-tert-butyl-2-methylcyclohexane.
¹H NMR Spectroscopy
The ¹H NMR spectrum is particularly informative. The distinct chemical shifts of the axial and equatorial protons on the cyclohexane ring, along with the signals from the methyl and tert-butyl groups, provide valuable information about the molecule’s structure and conformation. Careful analysis of coupling constants (J values) can further confirm the axial and equatorial assignments.
¹³C NMR Spectroscopy
¹³C NMR provides information about the carbon atoms in the molecule. Distinct chemical shifts for the carbons bearing the tert-butyl and methyl groups, as well as for the other ring carbons, aid in structural elucidation.
IR Spectroscopy
Infrared (IR) spectroscopy can reveal the presence of characteristic functional groups, such as the C-H stretches associated with the alkyl groups.
Mass Spectrometry
Mass spectrometry provides information about the molecular weight and fragmentation pattern, confirming the molecular formula.
Synthesis and Potential Applications
While the detailed synthesis pathways for cis-1-tert-butyl-2-methylcyclohexane are complex and require specialized knowledge of organic chemistry, its existence highlights the possibilities within organic synthesis and the challenges in controlling stereochemistry.
Synthetic Challenges
Precise control over stereochemistry is crucial in organic synthesis. The synthesis of this cis isomer would likely involve steps to ensure that the tert-butyl and methyl groups are introduced on the same side of the cyclohexane ring. This might involve selective reactions and potentially the use of chiral catalysts or reagents.
Potential Applications
Due to its relatively simple structure and the availability of its constituent groups, cis-1-tert-butyl-2-methylcyclohexane might find niche applications within organic chemistry research. For example, it could serve as a model system to study steric effects in cyclohexane derivatives or be used as a building block in the synthesis of more complex molecules. However, widespread industrial applications are less likely given its complex synthesis and lack of pronounced functional groups suitable for widespread industrial reactions.
Comparison with the Trans Isomer
Comparing cis-1-tert-butyl-2-methylcyclohexane with its trans isomer reveals the significant influence of stereochemistry on molecular properties. The trans isomer would exhibit a different conformational preference, with both the tert-butyl and methyl groups likely occupying equatorial positions in the most stable conformation, leading to a much lower degree of steric strain compared to the cis isomer. This difference in steric strain would manifest in differences in melting points, boiling points, and reactivity. The NMR spectra would also show distinct differences reflecting the different conformational preferences.
Conclusion
Cis-1-tert-butyl-2-methylcyclohexane, despite its seemingly simple structure, offers a rich case study in stereochemistry and conformational analysis. The interplay between the bulky tert-butyl group and the methyl group, along with the inherent flexibility of the cyclohexane ring, creates a molecule with fascinating properties dictated largely by its conformational preference. Understanding these intricacies requires a combined approach, employing both theoretical analysis and experimental techniques such as NMR spectroscopy. Although not currently found in widespread industrial applications, its study contributes significantly to the fundamental understanding of organic chemistry and its principles. Further research, including computational studies and potential novel synthetic pathways, may reveal new insights into its properties and potential applications in the future.
Latest Posts
Latest Posts
-
Which Of The Following Statements Are True About Proteins
Apr 03, 2025
-
Are Men And Women Treated Equally Essay
Apr 03, 2025
-
1 Km 2 To M 2
Apr 03, 2025
-
Carbon Is Good Conductor Of Electricity
Apr 03, 2025
-
Figure Abcd Is A Kite Find The Value Of X
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Cis 1 Tert Butyl 2 Methylcyclohexane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
