Boiling Point Of Sugar And Water
News Leon
Apr 04, 2025 · 5 min read
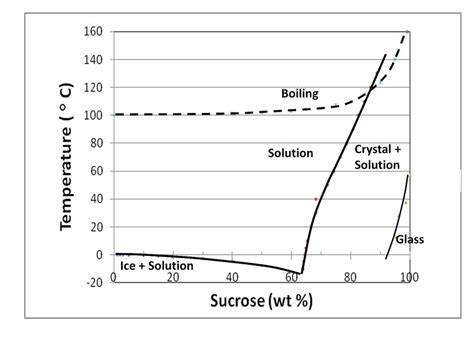
Table of Contents
Boiling Point of Sugar and Water: A Deep Dive
The seemingly simple question of what happens when you boil a sugar and water solution opens a fascinating door into the world of chemistry, specifically colligative properties. Understanding the boiling point elevation caused by adding sugar to water requires exploring concepts like vapor pressure, intermolecular forces, and the impact of solute concentration. This comprehensive guide will delve into these aspects, providing a detailed explanation of the boiling point of sugar-water solutions and related phenomena.
What is the Boiling Point of Pure Water?
Before we explore the complexities of sugar solutions, let's establish a baseline. Pure water boils at 100°C (212°F) at standard atmospheric pressure (1 atmosphere or 760 mmHg). This temperature represents the point at which the vapor pressure of water equals the atmospheric pressure, allowing water molecules to escape the liquid phase and transition into the gaseous phase (steam).
Factors Affecting Boiling Point of Water:
While 100°C is a commonly accepted value, several factors can subtly influence the boiling point of water:
-
Altitude: At higher altitudes, atmospheric pressure is lower. This means the water needs less energy to reach a vapor pressure equal to the surrounding pressure, resulting in a lower boiling point.
-
Impurities: The presence of dissolved substances, even in small amounts, can slightly elevate the boiling point. This is because the solute molecules interfere with the escape of water molecules from the surface.
-
Pressure: Increasing the external pressure on the water increases the boiling point. Conversely, decreasing the pressure lowers the boiling point. Pressure cookers utilize this principle to achieve higher cooking temperatures and faster cooking times.
The Impact of Sugar on Water's Boiling Point: Boiling Point Elevation
Adding sugar (sucrose) to water is a classic example of boiling point elevation. This phenomenon, a colligative property, depends solely on the number of solute particles (in this case, sugar molecules) present in the solution, not their identity. Sugar molecules, when dissolved, disrupt the hydrogen bonding network within the water. This disruption makes it more difficult for water molecules to escape into the gaseous phase, thus requiring a higher temperature to reach the boiling point.
Understanding Colligative Properties:
Colligative properties are characteristics of solutions that depend on the concentration of solute particles rather than their identity. Other colligative properties include:
- Freezing point depression: The freezing point of a solution is lower than that of the pure solvent.
- Vapor pressure lowering: The vapor pressure of a solution is lower than that of the pure solvent.
- Osmotic pressure: The pressure required to prevent the flow of solvent across a semipermeable membrane.
Calculating the Boiling Point Elevation of Sugar Water Solutions:
Precisely calculating the boiling point elevation of a sugar-water solution involves using the following formula:
ΔTb = Kb * m * i
Where:
- ΔTb is the boiling point elevation (the difference between the boiling point of the solution and the boiling point of the pure solvent).
- Kb is the ebullioscopic constant (a constant specific to the solvent – for water, Kb ≈ 0.512 °C/m).
- m is the molality of the solution (moles of solute per kilogram of solvent).
- i is the van't Hoff factor (a measure of the number of particles a solute dissociates into in solution – for sucrose, i ≈ 1 as it doesn't dissociate).
Example Calculation:
Let's calculate the boiling point elevation for a solution containing 100 grams of sucrose (C₁₂H₂₂O₁₁) dissolved in 1 kilogram of water.
-
Calculate the moles of sucrose: The molar mass of sucrose is approximately 342.3 g/mol. Therefore, 100g of sucrose is (100g)/(342.3 g/mol) ≈ 0.292 moles.
-
Calculate the molality: The molality is 0.292 moles / 1 kg water = 0.292 m.
-
Calculate the boiling point elevation: ΔTb = (0.512 °C/m) * (0.292 m) * (1) ≈ 0.15 °C
-
Determine the boiling point of the solution: The boiling point of the solution will be approximately 100.15°C.
Factors Affecting the Accuracy of Boiling Point Elevation Calculations:
The formula provided offers a good approximation, but several factors can affect the accuracy of the calculation:
-
Ideal vs. Non-Ideal Solutions: The formula assumes an ideal solution, where solute-solute and solute-solvent interactions are negligible. In reality, deviations from ideality can occur, especially at higher concentrations.
-
Ionic vs. Non-Ionic Solutes: The van't Hoff factor (i) is crucial for ionic compounds that dissociate into ions in solution. Sucrose, being a non-ionic compound, has an i value of approximately 1.
-
Association of Molecules: In some cases, solute molecules may associate in solution, affecting the effective number of particles and thus the boiling point elevation.
Practical Applications: Sugar and Water Boiling in Cooking and Food Science
The boiling point elevation of sugar-water solutions is highly relevant in several applications:
-
Candy Making: The production of various candies, such as caramel and fondant, relies on controlling the boiling point of sugar syrups to achieve the desired consistency. Higher sugar concentrations lead to higher boiling points, allowing for the creation of different candy textures.
-
Food Preservation: Boiling sugar solutions at higher temperatures can aid in preserving food by killing microorganisms.
-
Syrup Production: The manufacturing of various syrups, like maple syrup or corn syrup, involves precise control of the sugar concentration to determine the final boiling point and syrup consistency.
Beyond Boiling Point: Other Considerations When Heating Sugar-Water Solutions
It's vital to remember that heating sugar solutions can lead to further chemical changes beyond just boiling point elevation:
-
Caramelization: At higher temperatures, sucrose can undergo caramelization, a complex series of reactions that produce a brown color and characteristic flavor. This process occurs above the boiling point of the solution.
-
Decomposition: At extremely high temperatures, sucrose can decompose, producing undesirable byproducts.
-
Crystallization: Careful control is needed to prevent sugar crystallization, which can significantly alter the texture of the final product.
Conclusion:
Understanding the boiling point of sugar and water solutions is more than just a simple scientific concept. It forms the basis for understanding colligative properties and has crucial applications in various fields, particularly in cooking and food science. While a simple formula can provide an approximation, factors like solution ideality and potential chemical reactions need to be considered for accurate predictions and successful applications. By grasping these concepts, one can better control the properties of sugar-water mixtures and achieve desired results in culinary and other endeavors. The seemingly straightforward act of boiling sugar and water unveils a world of complex interactions and phenomena waiting to be explored.
Latest Posts
Latest Posts
-
What Is The Lightest Subatomic Particle
Apr 05, 2025
-
Which Valve Prevents Backflow Into The Left Ventricle
Apr 05, 2025
-
Cervical Vertebrae Differ From Other Vertebrae In What Way
Apr 05, 2025
-
What Is The Greatest Negative Integer
Apr 05, 2025
-
The Energy That Drives The Water Cycle Comes From The
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Boiling Point Of Sugar And Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
