1 12 The Mass Of A Carbon 12 Atom
News Leon
Apr 05, 2025 · 6 min read
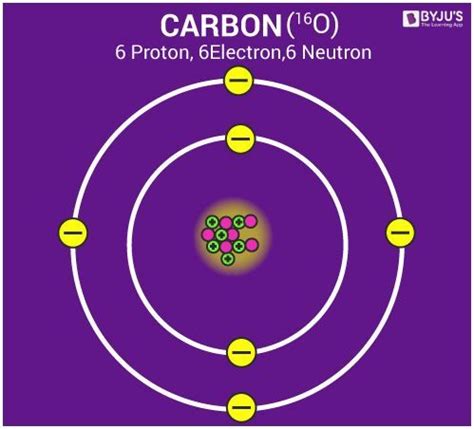
Table of Contents
1/12 the Mass of a Carbon-12 Atom: Unveiling the Foundation of Atomic Mass
The seemingly simple fraction, 1/12 the mass of a carbon-12 atom, forms the bedrock of our understanding of atomic mass. This seemingly arbitrary choice has profound implications for chemistry, physics, and numerous scientific disciplines. This article delves deep into the significance of this definition, exploring its historical context, scientific rationale, and its continuing relevance in modern science.
The Historical Context: A Need for Standardization
Before the adoption of the carbon-12 standard, the measurement of atomic mass was a chaotic landscape. Different scientists used different reference points, leading to inconsistencies and confusion in calculations and comparisons. Elements were assigned atomic weights relative to oxygen, hydrogen, or other elements, resulting in a confusing array of values. This lack of standardization hampered scientific progress and made collaborative research challenging.
The Oxygen Standard: A Precursor to Carbon-12
For a significant period, oxygen served as the standard for atomic mass. However, there was an ambiguity: two isotopes of oxygen existed in naturally occurring samples, leading to two distinct scales – one based on O-16 and another on the natural isotopic mix of oxygen. This duality fueled discrepancies in reported atomic masses, further highlighting the urgent need for a unified and precise standard.
The Advent of Carbon-12: A Solution to the Chaos
The International Union of Pure and Applied Chemistry (IUPAC) and the International Union of Pure and Applied Physics (IUPAP) recognized the limitations of the oxygen standard and the crucial need for a universally accepted reference point. In 1961, a landmark decision was made: carbon-12, an isotope of carbon with six protons and six neutrons, was chosen as the new standard.
This choice wasn't arbitrary. Carbon-12 offered several advantages:
-
Isotopic Purity: Unlike oxygen, carbon-12 is a single isotope, eliminating the ambiguity and inconsistencies associated with naturally occurring isotopic mixtures. This ensured a clear and consistent standard for all measurements.
-
Abundance and Availability: Carbon-12 is relatively abundant and readily available, making it practical for widespread use in mass spectrometry and other analytical techniques.
-
Well-Defined Properties: The properties of carbon-12 are well-understood and readily measurable, facilitating precise and reproducible measurements of atomic mass.
Defining the Atomic Mass Unit (amu): A Consequence of the Carbon-12 Standard
The adoption of carbon-12 as the standard led to the definition of the atomic mass unit (amu), also known as the dalton (Da). One atomic mass unit is defined as exactly 1/12 the mass of a single carbon-12 atom. This means that the mass of a carbon-12 atom is precisely 12 amu.
This seemingly simple definition has far-reaching consequences. All other atomic masses are now determined relative to the mass of carbon-12. For example, if an atom has twice the mass of a carbon-12 atom, its atomic mass is 24 amu. This standardization eliminates the previous inconsistencies and allows for accurate comparisons and calculations across different elements and isotopes.
The Significance of Precise Atomic Mass Measurement
Accurate determination of atomic mass is crucial for various scientific and technological applications:
1. Chemistry: Stoichiometry and Chemical Reactions
In chemistry, atomic mass is fundamental for stoichiometric calculations. It allows chemists to determine the relative amounts of reactants and products in chemical reactions, ensuring accurate predictions of reaction yields and compositions.
2. Nuclear Physics: Understanding Isotopes and Nuclear Reactions
Nuclear physics relies heavily on precise atomic mass measurements. The difference in mass between reactants and products in nuclear reactions reflects the energy released or absorbed during the process (Einstein's famous E=mc²). Understanding these mass differences is crucial for studying nuclear fission, fusion, and radioactive decay.
3. Mass Spectrometry: Identifying and Quantifying Molecules
Mass spectrometry is a powerful analytical technique used to identify and quantify different molecules based on their mass-to-charge ratio. The accuracy of mass spectrometry measurements hinges on the precise definition of the atomic mass unit, based on the carbon-12 standard.
4. Biochemistry: Understanding Macromolecules and Biological Processes
In biochemistry, the accurate measurement of molecular masses is critical for studying proteins, nucleic acids, and other macromolecules. Determining the precise mass of these molecules helps researchers to understand their structure, function, and their roles in biological processes.
5. Materials Science: Characterizing Materials and Properties
The composition and properties of materials are directly related to the atomic masses of their constituent elements. Precise atomic mass measurements are essential for developing new materials with specific characteristics and for quality control in materials science.
Beyond the Basics: Isotopic Abundance and Average Atomic Mass
While carbon-12 serves as the standard, naturally occurring carbon contains a small percentage of carbon-13. Therefore, the average atomic mass of carbon found in nature is slightly greater than 12 amu. This concept of average atomic mass takes into account the relative abundance of each isotope of an element. The average atomic mass is a weighted average of the masses of each isotope, considering their natural abundance.
This distinction between the standard atomic mass of carbon-12 and the average atomic mass of naturally occurring carbon highlights the importance of understanding both concepts in various scientific applications. While carbon-12 forms the basis for the atomic mass unit, the average atomic mass is frequently used in practical calculations involving naturally occurring samples.
The Ongoing Relevance of the Carbon-12 Standard
Despite the advancements in scientific techniques and our growing understanding of atomic structure, the carbon-12 standard remains the cornerstone of atomic mass measurement. Its simplicity, precision, and universal acceptance have cemented its place in the scientific community. The definition remains a testament to the power of international collaboration and the importance of standardized measurements in advancing scientific knowledge.
Future Directions: High-Precision Measurements and New Technologies
The ongoing refinement of measurement techniques continues to improve the precision of atomic mass determinations. Advanced mass spectrometry and other analytical methods are pushing the boundaries of accuracy, allowing for increasingly precise measurements of atomic masses and isotopic abundances. These advancements have implications for various scientific fields, including fundamental physics, nuclear chemistry, and materials science.
Furthermore, new technologies and approaches are constantly emerging that contribute to our understanding of atomic mass. For instance, advancements in computational chemistry and theoretical modeling are providing increasingly accurate predictions of atomic masses, complementing and validating experimental measurements.
Conclusion: A Cornerstone of Modern Science
In conclusion, 1/12 the mass of a carbon-12 atom is more than just a simple fraction; it is the foundation upon which our understanding of atomic mass rests. This seemingly arbitrary choice has had a profound and lasting impact on scientific progress, enabling precise measurements, standardized calculations, and collaborative research across diverse disciplines. As scientific techniques continue to advance, the carbon-12 standard will undoubtedly remain a crucial element in our ongoing quest to understand the fundamental building blocks of matter. Its enduring relevance underscores the significance of international cooperation and the pursuit of precise and universally accepted scientific standards. The impact of this single definition extends far beyond the laboratory, shaping our understanding of the universe at its most fundamental level.
Latest Posts
Latest Posts
-
The Pigment Molecules Responsible For Photosynthesis Are Located In The
Apr 05, 2025
-
Is The Paramecium A Unicellular Or Multicellular
Apr 05, 2025
-
Which Of The Following Is Not A Vector
Apr 05, 2025
-
Can 2 Sperm Enter One Egg
Apr 05, 2025
-
If A Ball Is Thrown Vertically Upward
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about 1 12 The Mass Of A Carbon 12 Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
