Why Is Ice Melting Not A Chemical Reaction
News Leon
Apr 02, 2025 · 5 min read
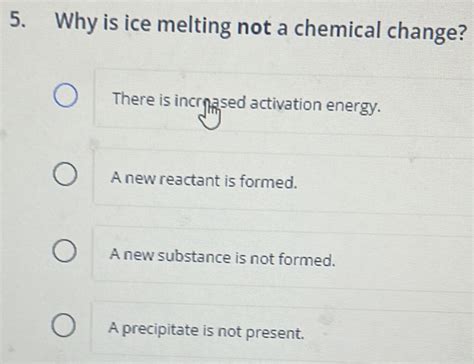
Table of Contents
Why Ice Melting Isn't a Chemical Reaction: A Deep Dive into Physical Changes
The simple act of ice melting—a ubiquitous process we observe daily—often sparks curiosity, especially regarding its classification as a physical change rather than a chemical reaction. While seemingly straightforward, understanding the nuanced difference between physical and chemical changes requires a closer examination of the underlying principles governing matter and its transformations. This comprehensive article delves into the reasons why ice melting is definitively a physical change, not a chemical reaction, exploring the concepts of phase transitions, molecular structure, and the conservation of matter.
Understanding the Core Differences: Physical Change vs. Chemical Reaction
Before dissecting the melting of ice, it’s crucial to establish a clear understanding of the fundamental differences between physical changes and chemical reactions. These two types of transformations represent distinct ways matter can alter its form and properties.
Physical Changes: These involve alterations in the physical properties of a substance, such as shape, size, state of matter (solid, liquid, gas), and density. Crucially, the chemical composition of the substance remains unchanged. Think of bending a paperclip, dissolving sugar in water, or even boiling water—the fundamental molecular structure of the substance stays the same. The change is purely physical, affecting only its outward appearance or arrangement.
Chemical Reactions: In contrast, chemical reactions involve a fundamental change in the chemical composition of a substance. New substances with different properties are formed, often involving the breaking and forming of chemical bonds. Burning wood, rusting iron, and baking a cake are classic examples of chemical reactions. The initial substances are transformed into entirely new ones with different chemical formulas.
The Melting of Ice: A Case Study in Physical Change
Ice, the solid form of water (H₂O), undergoes a phase transition when it melts. This transition involves a shift from a rigid, crystalline structure (solid) to a more fluid, mobile state (liquid). Let's examine this process through the lens of the key characteristics differentiating physical changes from chemical reactions:
1. No Change in Chemical Composition: The H₂O Remains Intact
The most critical aspect to understand is that when ice melts, the chemical formula of water remains unchanged. The molecules of H₂O, composed of two hydrogen atoms covalently bonded to one oxygen atom, retain their integrity throughout the entire process. There is no breaking or forming of chemical bonds. The transformation merely involves a change in the arrangement and interaction of these existing H₂O molecules.
2. Reversible Process: Freezing and Melting
Melting ice is a reversible process. By lowering the temperature below 0°C (32°F), the liquid water can be easily converted back into solid ice. This reversibility is a hallmark of physical changes. Chemical reactions, however, are often irreversible, or require significantly different conditions to reverse. You can't simply reverse the burning of wood to get the original wood back.
3. Energy Changes: Absorbing Heat, Not Creating New Bonds
Melting ice requires the absorption of energy, typically in the form of heat. This energy input weakens the intermolecular forces (hydrogen bonds) holding the H₂O molecules in their fixed positions within the ice crystal lattice. The energy doesn't break covalent bonds within the H₂O molecule itself, but instead overcomes the attractive forces between molecules, allowing them to move more freely. This is in stark contrast to chemical reactions where energy changes often accompany the making and breaking of chemical bonds.
4. No New Substances Formed: Still Just Water
Throughout the melting process, only water is present. There is no formation of new chemical compounds or elements. The liquid water retains all the chemical properties of the original ice, such as its ability to dissolve certain substances, its boiling point, and its specific heat capacity. The phase transition doesn't alter these fundamental chemical characteristics.
Molecular Perspective: Hydrogen Bonds and Phase Transitions
Delving into the molecular level further solidifies the argument for ice melting being a physical change. Water molecules are held together by hydrogen bonds – a special type of intermolecular force that is relatively weak compared to covalent bonds.
In ice, these hydrogen bonds create a highly ordered, crystalline structure. The molecules are arranged in a specific three-dimensional lattice, maximizing the hydrogen bonding and resulting in a relatively low density.
When heat is added, the energy disrupts these hydrogen bonds, allowing the water molecules to move more freely. The rigid structure of the ice lattice collapses, transitioning to the less ordered, more fluid structure of liquid water. Again, no covalent bonds within the H₂O molecules themselves are broken or formed. The change is purely in the arrangement and interaction of the water molecules, resulting in a change of state.
Addressing Common Misconceptions
Several misconceptions can cloud the understanding of why ice melting is a physical change:
- Appearance Change: While the transformation from solid ice to liquid water involves a significant change in appearance (from a rigid structure to a flowing liquid), this visual change alone doesn't signify a chemical reaction. Many physical changes result in noticeable alterations in appearance.
- Temperature Change: The temperature change during melting (0°C at standard pressure) is a consequence of the energy absorbed to overcome the intermolecular forces, not a characteristic of a chemical reaction. Many physical processes involve temperature changes.
- Energy Absorption: While energy is absorbed during the melting process, this energy input is used to overcome intermolecular forces, not to break chemical bonds. Energy changes are common in both physical and chemical processes.
Conclusion: The Irrefutable Case for a Physical Change
In conclusion, the melting of ice unequivocally represents a physical change, not a chemical reaction. This assertion is grounded in the following irrefutable evidence: the chemical composition of water (H₂O) remains unchanged, the process is reversible, energy is absorbed to overcome intermolecular forces, and no new substances are formed. Understanding this distinction is crucial for grasping fundamental concepts in chemistry and physics, emphasizing the importance of differentiating between alterations in the physical properties of a substance and changes in its fundamental chemical nature. The transformation from solid ice to liquid water serves as a perfect example of a phase transition driven by changes in intermolecular interactions, not by the creation or destruction of chemical bonds. This basic understanding lays the foundation for comprehending more complex physical and chemical processes.
Latest Posts
Latest Posts
-
Which Tissue Is Responsible For Plant Growth
Apr 03, 2025
-
Fill Up The Blanks With The Words Given
Apr 03, 2025
-
Which Of The Following Is True About Tsunamis
Apr 03, 2025
-
Which Of The Following Is Not A Transition Metal
Apr 03, 2025
-
When Air Is Heated What Happens
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Why Is Ice Melting Not A Chemical Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
