Which Subshell Is Represented By The Actinides Series
News Leon
Apr 03, 2025 · 5 min read
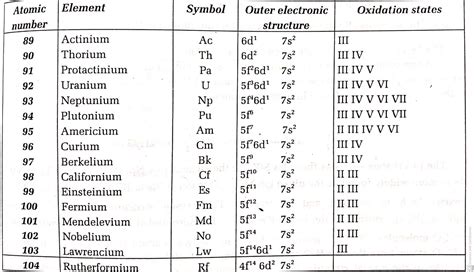
Table of Contents
Which Subshell is Represented by the Actinide Series? Understanding the f-Block Elements
The actinide series, a fascinating group of elements, holds a unique position in the periodic table. Understanding their location and electronic configuration is key to grasping their chemical behavior and remarkable properties. This article delves deep into the electronic structure of actinides, definitively answering the question: which subshell is represented by the actinides series? We'll explore the nuances of electron filling, the complexities of their chemistry, and the implications of their unique position in the periodic table.
The f-Block: Home of the Actinides
The answer is straightforward: the actinide series is represented by the 5f subshell. This placement defines their chemical behavior and distinguishes them from other element groups. Unlike the s-block (alkali and alkaline earth metals) and p-block (many nonmetals and metalloids), the actinides belong to the f-block, a group of elements characterized by the filling of the f atomic orbitals.
Understanding Atomic Orbitals and Electron Configuration
To fully understand the actinide series' location, it's crucial to grasp the basics of atomic orbitals. Electrons occupy specific regions of space around the nucleus called orbitals. These orbitals are categorized into different subshells: s, p, d, and f. The subshells can hold a varying number of electrons:
- s subshell: holds a maximum of 2 electrons
- p subshell: holds a maximum of 6 electrons
- d subshell: holds a maximum of 10 electrons
- f subshell: holds a maximum of 14 electrons
The filling of these subshells follows the Aufbau principle, which dictates that electrons occupy the lowest energy levels first. However, exceptions exist due to the complex interactions between electrons and the relative energies of orbitals.
The Aufbau Principle and Actinide Electron Configuration
The Aufbau principle generally predicts electron configurations. However, for the actinides, this principle, while providing a good starting point, doesn't perfectly describe the electronic structure of all actinide ions. The energies of the 5f and 6d orbitals are very close, leading to variations in electron configurations across the series.
For example, while thorium (Th) might be expected to have a simple [Rn] 5f² 6d² configuration, the actual configuration is often described as [Rn] 6d² 7s². This close energy proximity between the 5f and 6d orbitals leads to complex and sometimes unpredictable electron configurations across the actinide series. This is a key factor contributing to the complex and varied chemistry of these elements.
The Periodic Table and f-block Placement
The periodic table's arrangement reflects the electron configuration of elements. The actinide series occupies the bottom row of the periodic table, separate from the main body to maintain the table's structure and readability. This separation highlights the fact that the actinides are filling the 5f subshell, which isn't readily incorporated into the main body of the table without compromising its clarity.
The Chemistry of the Actinides: A Consequence of the 5f Subshell
The 5f electrons significantly influence the actinides' chemical properties. Several factors contribute to this:
-
Variable Oxidation States: Unlike many other elements with more predictable oxidation states, actinides exhibit a wide range of oxidation states. This is a direct consequence of the relatively similar energies of the 5f, 6d, and 7s orbitals. The ease with which electrons can be removed from these orbitals leads to this extensive variability. For example, uranium (U) can exist in oxidation states ranging from +3 to +6.
-
Complex Chemistry: The partially filled 5f orbitals lead to complex coordination chemistry. Actinides readily form coordination complexes with various ligands (molecules or ions that bind to a central metal atom). The geometry and bonding in these complexes are often highly intricate, further showcasing the unique properties of the 5f electrons.
-
Radioactivity: Most actinides are radioactive, meaning their nuclei are unstable and decay over time, emitting radiation. This radioactivity is a consequence of their relatively large and unstable nuclei. This radioactivity is a significant factor to consider when handling these elements and understanding their applications.
-
Relativistic Effects: The high atomic numbers of the actinides lead to significant relativistic effects. These effects alter the energies and sizes of the orbitals, influencing the chemical and physical properties of these elements. The relativistic contraction of the 6s and 6p orbitals, for instance, has an effect on the chemical bonding capabilities of the actinides.
Distinguishing Actinides from Lanthanides
Both the actinides and lanthanides are f-block elements, but they differ in several crucial aspects:
-
Subshell Filled: Lanthanides fill the 4f subshell, while actinides fill the 5f subshell. This seemingly minor difference in principal quantum number leads to significant variations in their chemical and physical properties.
-
Radioactivity: While some lanthanides exhibit weak radioactivity, the majority of actinides are significantly radioactive. This radioactivity is a key differentiator and presents major challenges in their handling and applications.
-
Oxidation States: While lanthanides primarily exhibit +3 oxidation states, actinides demonstrate a much broader range of oxidation states, adding to their chemical complexity.
-
Reactivity: Generally, actinides are more reactive than lanthanides, exhibiting greater tendencies to form compounds with a wider range of elements.
Applications of the Actinides
Despite their radioactivity and handling complexities, actinides find applications in various fields:
-
Nuclear Fuel: Uranium and plutonium are crucial in nuclear power generation. Their ability to undergo nuclear fission releases enormous amounts of energy, harnessable for electricity production.
-
Medical Applications: Some actinide isotopes are used in medical applications, although their radioactivity necessitates careful handling and administration. Targeted alpha therapy uses alpha-emitting isotopes to combat cancerous cells, utilizing the high energy of alpha particles.
-
Research: Actinides are invaluable in scientific research, particularly in nuclear physics and chemistry. Studying their properties helps us to understand nuclear processes, and their complex chemical behavior continues to challenge and stimulate researchers globally.
Conclusion: The 5f Subshell and its Significance
In conclusion, the actinide series is unequivocally represented by the 5f subshell. This placement is not merely a matter of orbital filling; it's the fundamental driver behind their unique and often complex chemistry, radioactivity, and diverse applications. Understanding the intricacies of the 5f electrons, their interactions with other orbitals, and the relativistic effects is essential for comprehending the fascinating and important world of actinide chemistry. Further research into these elements continues to expand our understanding of nuclear processes and the behavior of matter at its most fundamental level. The 5f subshell, therefore, represents more than just a location on the periodic table; it's a gateway to a universe of scientific exploration and technological advancement.
Latest Posts
Latest Posts
-
Which Of The Following Is An Antibiotic
Apr 04, 2025
-
Which Of The Following Statements About Friction Is True
Apr 04, 2025
-
A Number Is Divisible By 9 If
Apr 04, 2025
-
The Final Product Of Gene Expression Is
Apr 04, 2025
-
The Main Function Of Trna Is To
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Subshell Is Represented By The Actinides Series . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
