Which Of The Following Elements Is Most Electronegative
News Leon
Apr 01, 2025 · 5 min read
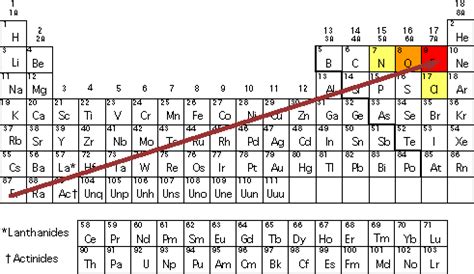
Table of Contents
Which of the Following Elements is Most Electronegative? A Deep Dive into Electronegativity
Electronegativity, a fundamental concept in chemistry, dictates how strongly an atom attracts electrons within a chemical bond. Understanding electronegativity is crucial for predicting the polarity of bonds, the geometry of molecules, and the reactivity of compounds. This article delves into the intricacies of electronegativity, exploring its trends in the periodic table and ultimately answering the question: which element reigns supreme in electron attraction? We'll explore various scales used to measure electronegativity, examine the factors influencing it, and highlight the significance of this property in chemical bonding and reactivity.
Understanding Electronegativity: A Definition and its Significance
Electronegativity quantifies the tendency of an atom to attract a shared pair of electrons within a chemical bond. It's not a directly measurable property like mass or charge, but rather a relative value that compares the electron-attracting power of different atoms. The higher the electronegativity value, the stronger the atom's pull on electrons.
Why is Electronegativity Important?
The significance of electronegativity extends across numerous chemical concepts:
-
Bond Polarity: The difference in electronegativity between two bonded atoms determines the polarity of the bond. A large difference leads to a polar covalent bond, where electrons are unequally shared, creating partial positive (δ+) and partial negative (δ-) charges. A very large difference results in an ionic bond, where electrons are essentially transferred from one atom to another.
-
Molecular Polarity: The overall polarity of a molecule depends on the polarities of its individual bonds and the molecule's geometry. Electronegativity helps predict the molecular dipole moment, which influences intermolecular forces and physical properties like boiling point and solubility.
-
Reactivity: Electronegativity plays a key role in predicting the reactivity of chemical species. Atoms with high electronegativity tend to be strong oxidizing agents, readily accepting electrons. Conversely, atoms with low electronegativity often act as reducing agents, easily donating electrons.
-
Acid-Base Chemistry: Electronegativity influences the acidity and basicity of molecules. Atoms with high electronegativity can stabilize negative charges, making the corresponding molecule a stronger acid.
Electronegativity Scales: Pauling, Mulliken, and Allred-Rochow
Several scales have been developed to quantify electronegativity, each with its own methodology and advantages:
1. Pauling Scale: The Most Widely Used
The most common and widely used scale is the Pauling scale, developed by Linus Pauling. This scale is based on bond energies and is relative, assigning a value of 4.0 to fluorine, the most electronegative element. Other elements are assigned values relative to fluorine. The Pauling scale is empirical, meaning it's based on experimental data rather than theoretical calculations.
2. Mulliken Scale: An Electrostatic Approach
The Mulliken scale takes a different approach, defining electronegativity as the average of the ionization energy (energy required to remove an electron) and electron affinity (energy released when an electron is added). This scale provides an absolute measure of electronegativity, unlike the relative Pauling scale. However, electron affinities are not always easily measurable for all elements.
3. Allred-Rochow Scale: Considering Effective Nuclear Charge
The Allred-Rochow scale considers the effective nuclear charge experienced by valence electrons and the size of the atom. It calculates electronegativity based on the electrostatic attraction between the nucleus and valence electrons. This scale offers a more theoretical approach, incorporating factors that influence the electron-attracting power of the nucleus.
Trends in Electronegativity Across the Periodic Table
Electronegativity exhibits clear trends across the periodic table:
-
Increases Across a Period: As you move from left to right across a period (row), electronegativity generally increases. This is because the number of protons in the nucleus increases, increasing the positive charge and thus the attraction for electrons. The shielding effect of inner electrons remains relatively constant across a period.
-
Decreases Down a Group: As you move down a group (column), electronegativity generally decreases. This is due to the increasing atomic size. The valence electrons are further from the nucleus, experiencing a weaker attraction. Furthermore, the increased number of inner electrons provides greater shielding, reducing the effective nuclear charge experienced by the valence electrons.
Identifying the Most Electronegative Element
Based on the most widely accepted Pauling scale, fluorine (F) is the most electronegative element with a value of 4.0. Its small atomic size and high effective nuclear charge contribute to its strong attraction for electrons. Oxygen (O) is a close second, followed by nitrogen (N) and chlorine (Cl). These elements are all located in the upper right corner of the periodic table, reflecting the trends discussed above.
Factors Affecting Electronegativity
Several factors contribute to an element's electronegativity:
-
Nuclear Charge: A higher nuclear charge leads to a stronger attraction for electrons.
-
Atomic Radius: A smaller atomic radius results in a stronger attraction because the valence electrons are closer to the nucleus.
-
Shielding Effect: Inner electrons shield the valence electrons from the full positive charge of the nucleus, reducing the effective nuclear charge.
-
Electron Configuration: Elements with nearly filled valence shells tend to have higher electronegativities as they are more likely to attract additional electrons to complete their octet.
Applications of Electronegativity
Understanding electronegativity is crucial in various areas of chemistry and beyond:
-
Predicting Bond Types: Knowing the electronegativity difference between atoms allows for accurate prediction of bond type (ionic, covalent, polar covalent).
-
Molecular Geometry: Electronegativity influences the distribution of electron density within a molecule, impacting its geometry.
-
Chemical Reactivity: Electronegativity helps predict the reactivity of elements and compounds.
-
Drug Design: In medicinal chemistry, electronegativity is considered when designing drugs to interact with specific biological targets.
-
Materials Science: The properties of materials are often influenced by the electronegativity of their constituent atoms.
Conclusion: Fluorine's Reign Supreme
This comprehensive exploration of electronegativity demonstrates its fundamental importance in chemistry. While various scales exist for its measurement, the Pauling scale remains the most commonly used. The trends across the periodic table highlight the influence of nuclear charge, atomic radius, and shielding effects. Ultimately, fluorine stands out as the most electronegative element, possessing the strongest attraction for electrons due to its unique combination of properties. Understanding electronegativity is essential for predicting chemical behavior, designing new materials, and advancing our comprehension of the molecular world. Its impact extends far beyond the theoretical realm, influencing diverse fields and shaping our understanding of chemical interactions. The mastery of this concept is crucial for any aspiring chemist or anyone seeking a deeper understanding of the forces that govern the molecular universe.
Latest Posts
Latest Posts
-
Which Statement Is True About Bacteria
Apr 03, 2025
-
Why Are Fleas Hard To Squish
Apr 03, 2025
-
How Many Chromosomes In A Daughter Cell
Apr 03, 2025
-
Why Do Bones Heal Quicker Than Cartilage
Apr 03, 2025
-
Can Magnitude Be Negative In Physics
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Elements Is Most Electronegative . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
