Which Of The Following Carbohydrates Is The Largest
News Leon
Apr 05, 2025 · 4 min read
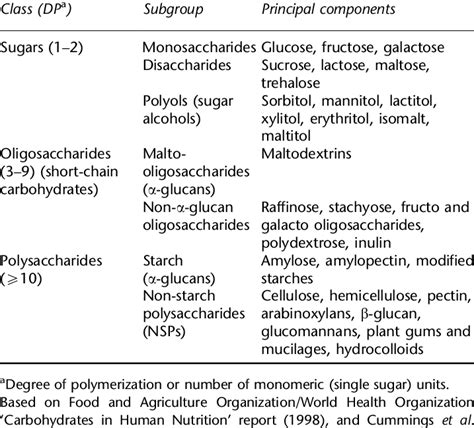
Table of Contents
Which of the Following Carbohydrates is the Largest? Understanding Macromolecule Size and Structure
Determining the "largest" carbohydrate among a group requires careful consideration. It's not simply a matter of comparing molecular weights, as carbohydrate structures are incredibly diverse and their size can be expressed in several ways. We need to understand the different types of carbohydrates, their polymerization, and how their structures affect their overall dimensions. This article will delve into the complexities of carbohydrate size, exploring different aspects to help you effectively compare and contrast various carbohydrate molecules.
Defining "Largest": Molecular Weight vs. Spatial Dimensions
Before we can even begin to compare the size of carbohydrates, we must define what we mean by "largest." There are two primary ways to interpret this:
1. Molecular Weight (MW): This is the sum of the atomic weights of all atoms in a carbohydrate molecule. Larger molecular weights generally indicate larger molecules. However, this isn't the whole story, as the same molecular weight can result in vastly different three-dimensional structures.
2. Spatial Dimensions: This considers the physical size and shape of the carbohydrate molecule in three-dimensional space. A branched carbohydrate might have a smaller molecular weight than a linear one, but its branched structure could result in a larger overall volume. Factors like branching, folding, and interactions with solvents significantly impact spatial dimensions.
Types of Carbohydrates and Their Potential for Size
Carbohydrates are broadly classified into three main types:
1. Monosaccharides: These are the simplest carbohydrates, the building blocks of larger carbohydrates. Examples include glucose, fructose, and galactose. Monosaccharides are relatively small molecules.
2. Oligosaccharides: These are short chains of monosaccharides linked together by glycosidic bonds. They typically contain 2-10 monosaccharide units. Examples include disaccharides like sucrose (glucose + fructose) and lactose (glucose + galactose), and trisaccharides like raffinose. Oligosaccharides are larger than monosaccharides but still relatively small compared to polysaccharides.
3. Polysaccharides: These are long chains of monosaccharides linked together by glycosidic bonds. They can contain hundreds or even thousands of monosaccharide units. Polysaccharides are the largest carbohydrates, exhibiting a wide range of sizes and structures. Examples include starch, glycogen, and cellulose.
Comparing Polysaccharide Size: Starch, Glycogen, and Cellulose
To illustrate the concept of size in carbohydrates, let's compare three common polysaccharides: starch, glycogen, and cellulose. All three are composed of glucose units, but their structures differ significantly, leading to variations in size and properties.
Starch: A Mixture of Amylose and Amylopectin
Starch is a storage polysaccharide found in plants. It's a mixture of two types of glucose polymers:
-
Amylose: This is a linear chain of glucose units linked by α-1,4-glycosidic bonds. Amylose molecules can be quite long, but they are still relatively unbranched.
-
Amylopectin: This is a branched-chain polymer of glucose units. It contains both α-1,4 and α-1,6 glycosidic bonds, creating branch points. Amylopectin molecules are significantly larger than amylose molecules due to their branching.
The overall size of starch granules depends on the ratio of amylose to amylopectin, as well as the length of the individual polymer chains.
Glycogen: The Animal Storage Polysaccharide
Glycogen is the storage polysaccharide found in animals. It's similar to amylopectin in structure, being a highly branched polymer of glucose units linked by α-1,4 and α-1,6 glycosidic bonds. However, glycogen is even more highly branched than amylopectin, resulting in a more compact structure. This highly branched nature allows for rapid mobilization of glucose units when needed for energy. Glycogen molecules are generally larger than amylopectin molecules due to their extensive branching.
Cellulose: The Structural Polysaccharide of Plants
Cellulose is a structural polysaccharide found in the cell walls of plants. It's a linear chain of glucose units linked by β-1,4-glycosidic bonds. The β-linkage results in a straight, rigid structure, allowing cellulose molecules to pack tightly together to form strong fibers. While individual cellulose chains can be very long, their linear nature means that their overall spatial dimensions might be comparable to, or even smaller than, the highly branched glycogen or amylopectin molecules, depending on the length of the chains.
Factors Affecting Carbohydrate Size
Several factors influence the overall size of a carbohydrate molecule:
-
Degree of Polymerization (DP): This refers to the number of monosaccharide units in a polysaccharide. A higher DP means a larger molecule.
-
Branching: Branched polysaccharides often have a larger overall volume than linear polysaccharides with the same DP.
-
Conformation: The three-dimensional shape of the molecule affects its size. Folding and coiling can make a molecule appear smaller than its extended length might suggest.
-
Interactions with Solvent: The interaction of the carbohydrate with the surrounding solvent (e.g., water) influences its conformation and thus its apparent size.
Conclusion: There's No Single "Largest" Carbohydrate
There is no single answer to the question of which carbohydrate is the largest. The answer depends on how you define "largest" – molecular weight or spatial dimensions – and on the specific carbohydrates being compared. Polysaccharides, particularly highly branched ones like glycogen, generally have the highest molecular weights and can occupy significant spatial volume. However, extremely long linear polysaccharides, like cellulose, may achieve considerable length despite a relatively smaller molecular weight compared to highly branched counterparts of similar length. Careful consideration of all these factors is necessary for a comprehensive understanding of carbohydrate size and structure. The specific molecular weight and spatial dimensions of a given carbohydrate will also vary depending on the source and conditions.
Latest Posts
Latest Posts
-
What Is The Smallest Unit Of Evolution
Apr 05, 2025
-
What Is The Percentage Of 40
Apr 05, 2025
-
Explain How We Perceive Objects As They Are
Apr 05, 2025
-
Which Of The Following Is A Natural Polymer
Apr 05, 2025
-
How Are All The Isotopes Of An Element Similar
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Carbohydrates Is The Largest . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
