Which Of The Following Are Properties Of A Metal
News Leon
Apr 05, 2025 · 6 min read
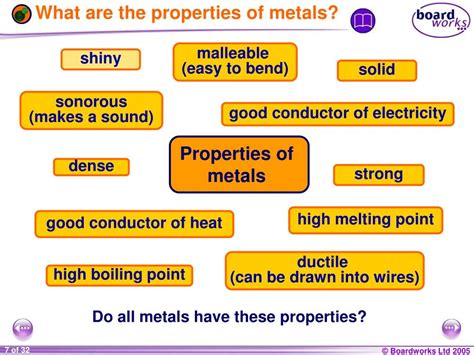
Table of Contents
Which of the Following are Properties of a Metal? A Deep Dive into Metallic Characteristics
Metals are ubiquitous in our lives, from the smartphones in our pockets to the skyscrapers that define our cities. Their widespread use stems from a unique set of properties that distinguish them from other classes of materials like nonmetals and metalloids. Understanding these properties is crucial in various fields, from material science and engineering to chemistry and physics. This article will delve into the defining characteristics of metals, exploring their physical and chemical properties in detail.
Physical Properties of Metals
Metals exhibit a range of physical properties that make them readily identifiable. These properties are largely determined by the metallic bonding that holds metal atoms together. Let's explore some of the key physical properties:
1. High Electrical Conductivity:
This is perhaps the most recognizable property of metals. Their excellent electrical conductivity is due to the delocalized electrons in the metallic lattice. These electrons are not bound to individual atoms but are free to move throughout the entire structure, allowing for the easy flow of electric current. This property is exploited in countless applications, from electrical wiring to electronic components. Silver and copper are particularly known for their high electrical conductivity.
2. High Thermal Conductivity:
Similar to electrical conductivity, metals also exhibit high thermal conductivity. The free-moving electrons efficiently transfer thermal energy throughout the metal, leading to rapid heat distribution. This is why metal pots and pans are commonly used for cooking – they heat up quickly and evenly. Aluminum is a prime example of a metal with high thermal conductivity, often used in heat sinks and cookware.
3. Malleability and Ductility:
These properties describe the ability of a metal to be deformed without breaking. Malleability refers to the ability to be hammered or rolled into sheets, while ductility refers to the ability to be drawn into wires. These properties are a direct consequence of the metallic bonding. The layers of atoms in a metal can slide over each other relatively easily without disrupting the overall structure. Gold is famously malleable and ductile, allowing it to be shaped into intricate designs.
4. Luster:
Metals typically have a characteristic metallic luster, meaning they have a shiny appearance. This is due to the interaction of light with the delocalized electrons in the metal lattice. The electrons absorb and re-emit light at various wavelengths, giving metals their characteristic sheen. This property is often used for decorative purposes, as seen in jewelry and other ornamental items.
5. High Density:
Most metals have a relatively high density compared to nonmetals. This is due to the close packing of atoms in the metallic lattice. The strong metallic bonds hold the atoms tightly together, resulting in a compact structure. Osmium and iridium are among the densest naturally occurring elements.
6. Hardness:
While not all metals are equally hard, many possess a significant degree of hardness. The strength of the metallic bonds contributes to the resistance to scratching and indentation. However, the hardness of a metal can be significantly affected by factors such as alloying and processing techniques. Steel, an alloy of iron and carbon, is known for its high hardness and strength.
7. Sonorousness:
Many metals produce a ringing sound when struck, a property known as sonorousness. This property is related to the elasticity of the metal and the way it vibrates when struck. The vibrations are transmitted through the metal lattice, producing sound waves. Bells, often made from bronze (an alloy of copper and tin), are a classic example of this property.
Chemical Properties of Metals
In addition to their physical properties, metals also exhibit characteristic chemical behaviors:
1. Reactivity:
Metals vary greatly in their reactivity, which is their tendency to undergo chemical reactions. Some metals, like alkali metals (e.g., sodium, potassium), are highly reactive and readily react with oxygen and water. Others, like noble metals (e.g., gold, platinum), are much less reactive and resistant to corrosion. The reactivity of a metal is influenced by its position in the periodic table and its electron configuration.
2. Oxidation:
Metals readily lose electrons to form positive ions (cations). This process is called oxidation. The ease with which a metal loses electrons determines its reactivity. Oxidation is a crucial process in many chemical reactions involving metals, including corrosion and the formation of metal oxides.
3. Corrosion:
Many metals undergo corrosion when exposed to the environment. Corrosion is essentially the deterioration of a metal due to chemical reactions with its surroundings. The most common form of corrosion is rusting, which is the oxidation of iron in the presence of oxygen and water. The formation of a protective oxide layer on some metals, like aluminum, can actually prevent further corrosion.
4. Formation of Alloys:
Metals can be combined with other metals or nonmetals to form alloys. Alloys possess properties that are often superior to those of the constituent metals. Steel, brass, and bronze are examples of commonly used alloys with enhanced strength, durability, or other desirable characteristics. The properties of an alloy depend on the composition and the interactions between the constituent elements.
Distinguishing Metals from Nonmetals and Metalloids
To fully grasp the properties of metals, it's essential to understand how they differ from other classes of materials:
Metals vs. Nonmetals: A Comparison
| Feature | Metals | Nonmetals |
|---|---|---|
| Electrical Conductivity | High | Low or Insulators |
| Thermal Conductivity | High | Low |
| Malleability/Ductility | High | Low (brittle) |
| Luster | Metallic luster | Variable, often dull |
| Density | Generally high | Generally low |
| Hardness | Variable, but often hard | Variable, often soft or brittle |
| Reactivity | Variable, some highly reactive | Variable, some highly reactive |
| Oxidation | Readily lose electrons to form cations | Gain electrons to form anions |
Metals vs. Metalloids: A Subtle Distinction
Metalloids, also known as semimetals, occupy an intermediate position between metals and nonmetals in the periodic table. They exhibit properties of both metals and nonmetals, making them useful in applications requiring unique combinations of characteristics. For instance, silicon, a metalloid, is a semiconductor, possessing intermediate electrical conductivity, making it crucial in electronic devices. Unlike metals, metalloids are generally brittle and lack metallic luster.
Conclusion
The properties of metals are a fascinating area of study, with implications across a vast range of disciplines. Their unique combination of physical and chemical properties – high electrical and thermal conductivity, malleability, ductility, luster, and their tendency to form positive ions – makes them indispensable in modern society. From the construction of buildings and bridges to the development of advanced technologies, the understanding and utilization of metallic properties continue to drive innovation and progress. Further research into new alloys and metal-based materials promises to unlock even greater potential for these remarkable substances.
Latest Posts
Latest Posts
-
Bangalore Is Capital Of Which State
Apr 06, 2025
-
Cytoplasm Divides Immediately After This Period
Apr 06, 2025
-
What Is The Percent Composition Of Co
Apr 06, 2025
-
In The Visible Spectrum Which Color Has The Longest Wavelength
Apr 06, 2025
-
Which Of The Following Statements About Stigma Is True
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Are Properties Of A Metal . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
