Which Elements Only Have One Valence Electron
News Leon
Mar 31, 2025 · 6 min read
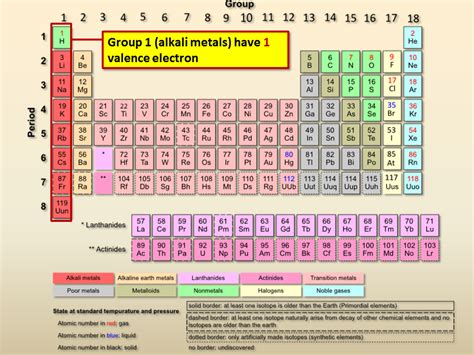
Table of Contents
- Which Elements Only Have One Valence Electron
- Table of Contents
- Which Elements Only Have One Valence Electron? Understanding Alkali Metals and Their Properties
- Identifying Elements with One Valence Electron: The Alkali Metals
- Understanding Valence Electrons and Their Significance
- Properties of Alkali Metals: A Detailed Exploration
- 1. Low Ionization Energies: The Ease of Electron Loss
- 2. Low Electronegativity: Unwillingness to Gain Electrons
- 3. Metallic Character: Conductivity and Malleability
- 4. Low Density: Light in Weight
- 5. Reactivity: Vigorous Reactions with Other Elements
- 6. Appearance: Silvery and Soft
- Chemical Reactions of Alkali Metals: A Closer Look
- 1. Reaction with Water: A Highly Exothermic Process
- 2. Reaction with Halogens: Formation of Ionic Compounds
- 3. Reaction with Oxygen: Formation of Oxides and Peroxides
- Applications of Alkali Metals: From Everyday Life to Advanced Technologies
- 1. Lithium: Batteries and Ceramics
- 2. Sodium: Sodium Lamps and Soaps
- 3. Potassium: Fertilizers and Medicine
- 4. Rubidium and Cesium: Atomic Clocks and Scientific Instruments
- 5. Francium: Limited Applications Due to Radioactivity
- Conclusion: The Unique Role of Alkali Metals
- Latest Posts
- Latest Posts
- Related Post
Which Elements Only Have One Valence Electron? Understanding Alkali Metals and Their Properties
The quest to understand the behavior of elements lies at the heart of chemistry. One crucial aspect of an element's properties is its valence electrons – the electrons in the outermost shell that participate in chemical bonding. Elements with only one valence electron exhibit unique characteristics, primarily driven by their strong tendency to lose that single electron and achieve a stable electron configuration. This article delves deep into these elements, focusing on their identification, properties, reactivity, and applications.
Identifying Elements with One Valence Electron: The Alkali Metals
The elements possessing only one valence electron are the alkali metals. This group, located in Group 1 (IA) of the periodic table, includes:
- Lithium (Li): Atomic number 3
- Sodium (Na): Atomic number 11
- Potassium (K): Atomic number 19
- Rubidium (Rb): Atomic number 37
- Cesium (Cs): Atomic number 55
- Francium (Fr): Atomic number 87
These elements share a common electronic configuration in their outermost shell, featuring a single s-electron. This single electron is relatively loosely held, making these elements highly reactive.
Understanding Valence Electrons and Their Significance
Before we delve deeper into the specifics of alkali metals, let's solidify our understanding of valence electrons. Valence electrons determine an element's chemical behavior. They are the electrons most likely to be involved in forming chemical bonds with other atoms. Elements strive to achieve a stable electron configuration, often resembling that of a noble gas (Group 18 elements). This stable configuration usually involves a full outermost electron shell, often eight electrons (the octet rule, though there are exceptions). Since alkali metals have only one valence electron, they readily lose this electron to achieve a stable, noble gas configuration.
Properties of Alkali Metals: A Detailed Exploration
The presence of only one valence electron significantly impacts the properties of alkali metals. Let's explore these properties in detail:
1. Low Ionization Energies: The Ease of Electron Loss
Alkali metals have exceptionally low ionization energies. This means that it takes very little energy to remove their single valence electron, forming a positively charged ion (cation). This low ionization energy is a direct consequence of the single, loosely held valence electron being far from the positively charged nucleus and shielded by inner electrons.
2. Low Electronegativity: Unwillingness to Gain Electrons
Alkali metals also possess low electronegativity. Electronegativity measures an atom's ability to attract electrons in a chemical bond. Since alkali metals readily lose their electron, they have a low affinity for gaining electrons. They are much more likely to donate their electron than to accept one.
3. Metallic Character: Conductivity and Malleability
Alkali metals exhibit strong metallic character. This means they are excellent conductors of heat and electricity. Their metallic bonding, where valence electrons are delocalized and shared amongst many atoms, facilitates the easy movement of electrons, leading to high conductivity. They are also malleable (easily shaped) and ductile (easily drawn into wires), typical properties of metals.
4. Low Density: Light in Weight
Compared to other metals, alkali metals have remarkably low densities. Lithium, in fact, is the least dense solid metal. This low density is related to their relatively large atomic radii and weak metallic bonding.
5. Reactivity: Vigorous Reactions with Other Elements
Perhaps the most striking characteristic of alkali metals is their high reactivity. Their strong tendency to lose their single valence electron makes them extremely reactive with various substances, particularly water and halogens. Reactions with water are often vigorous and exothermic (releasing heat), sometimes even explosive.
6. Appearance: Silvery and Soft
Alkali metals generally have a silvery-white appearance when freshly cut. They are also relatively soft and can be easily cut with a knife. However, exposure to air leads to rapid oxidation and tarnishing.
Chemical Reactions of Alkali Metals: A Closer Look
The reactivity of alkali metals stems from their eagerness to lose their single valence electron. Let’s examine some key reactions:
1. Reaction with Water: A Highly Exothermic Process
The reaction of alkali metals with water is a classic example of their high reactivity. The general reaction can be represented as:
2M(s) + 2H₂O(l) → 2MOH(aq) + H₂(g)
where M represents an alkali metal. The reaction produces a metal hydroxide (a strong base) and hydrogen gas. The reaction's vigor increases as you move down the group (from Li to Fr). Lithium reacts relatively slowly, while sodium reacts vigorously, potassium even more so, and rubidium and cesium react violently, often igniting the hydrogen gas produced.
2. Reaction with Halogens: Formation of Ionic Compounds
Alkali metals readily react with halogens (Group 17 elements) to form ionic compounds called halides. For example, sodium reacts with chlorine to form sodium chloride (NaCl), common table salt. The reaction involves the transfer of the valence electron from sodium to chlorine, resulting in the formation of Na⁺ and Cl⁻ ions, held together by electrostatic attraction.
3. Reaction with Oxygen: Formation of Oxides and Peroxides
The reaction of alkali metals with oxygen is more complex than their reactions with water or halogens. Lithium forms lithium oxide (Li₂O), while sodium forms sodium peroxide (Na₂O₂). Potassium, rubidium, and cesium form superoxides (e.g., KO₂). The formation of different oxides reflects the increasing reactivity down the group.
Applications of Alkali Metals: From Everyday Life to Advanced Technologies
Alkali metals, despite their high reactivity, find widespread applications in various fields:
1. Lithium: Batteries and Ceramics
Lithium's lightweight nature and high electrochemical potential make it crucial in lithium-ion batteries, powering portable electronic devices and electric vehicles. Lithium compounds are also used in ceramics and glass production.
2. Sodium: Sodium Lamps and Soaps
Sodium finds use in sodium-vapor lamps, providing efficient and bright street lighting. Sodium compounds are essential in the manufacture of soaps and detergents.
3. Potassium: Fertilizers and Medicine
Potassium is vital for plant growth, making potassium compounds crucial in fertilizers. Potassium salts also have applications in medicine, particularly in treating electrolyte imbalances.
4. Rubidium and Cesium: Atomic Clocks and Scientific Instruments
Rubidium and cesium have relatively low ionization energies, making them valuable in atomic clocks and other scientific instruments requiring precise measurements of time and frequency. Cesium is used in some photoelectric cells.
5. Francium: Limited Applications Due to Radioactivity
Francium, being highly radioactive and short-lived, has very limited practical applications, primarily confined to scientific research.
Conclusion: The Unique Role of Alkali Metals
Elements with only one valence electron, the alkali metals, are a fascinating group with distinctive properties driven by their single valence electron. Their high reactivity, low ionization energies, and low electronegativities make them essential in various applications, ranging from everyday products to cutting-edge technologies. Understanding their unique characteristics is crucial for appreciating their importance in chemistry and numerous technological advancements. Further research continues to unlock new applications and deepen our understanding of these remarkable elements.
Latest Posts
Latest Posts
-
What Is An Overview Of A Company
Apr 03, 2025
-
What Is 0 15 As A Percent
Apr 03, 2025
-
A Battery Consists Of Five Dry Cells Connected In Series
Apr 03, 2025
-
How To Write Permission Letter To Travel
Apr 03, 2025
-
A Solution With A Ph Of 5 0 Is
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Elements Only Have One Valence Electron . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
