What Type Of Bond Links Amino Acids Together
News Leon
Apr 03, 2025 · 6 min read
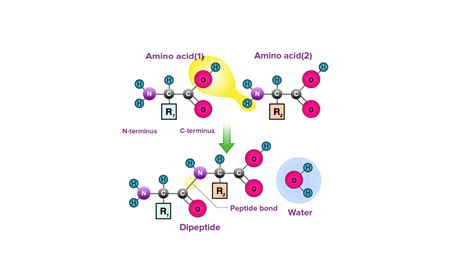
Table of Contents
What Type of Bond Links Amino Acids Together? Exploring Peptide Bonds in Proteins
The building blocks of life, proteins, are complex macromolecules crucial for virtually every biological process. Understanding their structure is paramount to understanding their function. At the heart of this understanding lies the peptide bond, the specific type of covalent bond that links amino acids together to form polypeptide chains, the precursors to proteins. This article delves deep into the nature of peptide bonds, exploring their formation, characteristics, and significance in protein structure and function.
The Amino Acid Foundation: Understanding the Monomers
Before we delve into the specifics of peptide bonds, let's briefly review the structure of amino acids, the individual units that come together to build proteins. An amino acid possesses a central carbon atom (the α-carbon) bonded to four distinct groups:
- An amino group (-NH₂): This group is basic and acts as a proton acceptor.
- A carboxyl group (-COOH): This group is acidic and acts as a proton donor.
- A hydrogen atom (-H): Simple and crucial for the overall structure.
- A side chain (R-group): This group is variable and determines the unique properties of each of the 20 standard amino acids. The R-group can be nonpolar, polar, acidic, or basic, significantly influencing the protein's overall structure and function.
The diversity of R-groups is what allows proteins to adopt a vast array of shapes and functions, contributing to the complexity of life.
Peptide Bond Formation: Dehydration Synthesis
The formation of a peptide bond is a condensation reaction, also known as a dehydration synthesis. This process involves the removal of a water molecule (H₂O) as the carboxyl group of one amino acid reacts with the amino group of another. Let's break down the steps:
-
Approximation: Two amino acids approach each other, positioning the carboxyl group of one amino acid close to the amino group of the other.
-
Reaction: The hydroxyl group (-OH) from the carboxyl group of the first amino acid and a hydrogen atom (-H) from the amino group of the second amino acid are released as a water molecule.
-
Bond Formation: A covalent bond forms between the carbon atom of the carboxyl group and the nitrogen atom of the amino group. This newly formed covalent bond is the peptide bond, also known as an amide bond.
The resulting molecule is a dipeptide, containing two amino acids linked by a peptide bond. This process can continue, adding more amino acids to the chain, forming a polypeptide. The sequence of amino acids in a polypeptide chain is known as the primary structure of the protein and is determined by the genetic code.
Characteristics of the Peptide Bond: Planarity and Partial Double Bond Character
The peptide bond possesses several crucial characteristics that influence protein structure:
-
Planarity: The peptide bond is essentially planar, meaning the six atoms involved in the bond (the carbon and oxygen of the carboxyl group, the nitrogen and hydrogen of the amino group, and the α-carbon atoms of the two amino acids) lie in the same plane. This planarity restricts rotation around the peptide bond.
-
Partial Double Bond Character: Due to resonance, the peptide bond exhibits partial double-bond character. This means that the electrons are delocalized between the carbon and nitrogen atoms, creating a resonance hybrid. This partial double bond character prevents free rotation around the peptide bond, further contributing to the rigidity of the polypeptide chain. However, rotation is permitted around the bonds adjacent to the peptide bond, specifically the α-carbon to carbonyl carbon bond (Phi - φ) and the α-carbon to amide nitrogen bond (Psi - ψ). This ability to rotate is crucial for the formation of secondary, tertiary, and quaternary protein structures.
-
Polarity: The peptide bond is polar due to the electronegativity difference between the oxygen and nitrogen atoms. This polarity contributes to the hydrogen bonding capabilities of the polypeptide chain, which are essential for the formation of secondary protein structures like alpha-helices and beta-sheets.
Peptide Bond Cleavage: Hydrolysis
The opposite of peptide bond formation is hydrolysis. This process breaks the peptide bond by adding a water molecule, resulting in the separation of amino acids. Hydrolysis can occur spontaneously under specific conditions, but it is often catalyzed by enzymes called proteases or peptidases. These enzymes play crucial roles in protein digestion, protein processing, and regulation of cellular processes.
The Significance of Peptide Bonds in Protein Structure and Function
The peptide bond's unique properties are fundamentally important for the overall structure and function of proteins:
-
Primary Structure: The sequence of amino acids linked by peptide bonds determines the primary structure of a protein. This sequence dictates the higher-order structures and ultimately the protein's function.
-
Secondary Structure: The hydrogen bonding between the peptide bond's polar carbonyl and amide groups contributes significantly to the formation of secondary structures, such as alpha-helices and beta-sheets. These structures are stabilized by hydrogen bonds between nearby amino acids within the polypeptide chain.
-
Tertiary Structure: The tertiary structure of a protein describes its three-dimensional folding pattern, which is driven by various interactions, including hydrophobic interactions, disulfide bonds, ionic bonds, and hydrogen bonds. The peptide bonds serve as the backbone for this intricate folding.
-
Quaternary Structure: Some proteins have a quaternary structure, comprising multiple polypeptide chains (subunits) interacting to form a functional protein complex. Peptide bonds form the backbone of each subunit, and interactions between subunits are crucial for the overall function of the protein complex.
Peptide Bond Modifications: Beyond the Basics
While the standard peptide bond is crucial, it's important to note that peptide bonds can undergo modifications that influence protein function:
-
Isopeptide bonds: These are unusual peptide bonds where the carboxyl group of one amino acid is linked to an atom other than the α-amino group of another amino acid. Examples include bonds to lysine side chains.
-
Post-translational modifications: After a protein is synthesized, various chemical groups can be added to amino acid side chains or the peptide backbone, altering its properties and function. These modifications can include glycosylation, phosphorylation, and ubiquitination.
-
Cyclic peptides: Some peptides form cyclic structures through the formation of additional covalent bonds between amino acid side chains or between the N- and C-termini. These cyclic peptides often exhibit enhanced stability and unique biological activities.
Conclusion: The Peptide Bond – A Cornerstone of Life
The peptide bond is far more than just a simple covalent bond; it is a cornerstone of biological structure and function. Its unique properties—planarity, partial double bond character, and polarity—dictate the folding patterns and biological activities of proteins. Understanding the intricacies of the peptide bond is essential for comprehending the vast complexity and diversity of proteins and their indispensable roles in life processes. From the simplest enzymes to the most complex molecular machines, the peptide bond remains the fundamental link in the chain of life. Further research continues to unveil the nuances of peptide bond modifications and their roles in disease and development, constantly expanding our understanding of this vital molecular connection.
Latest Posts
Latest Posts
-
Oxygen Released During Photosynthesis Comes From
Apr 04, 2025
-
Which Of The Following Defines A Coenzyme
Apr 04, 2025
-
When There Is A Decrease In Both Demand And Supply
Apr 04, 2025
-
Is A Euglena A Prokaryote Or Eukaryote
Apr 04, 2025
-
Containing Two Different Alleles For A Trait
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Type Of Bond Links Amino Acids Together . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
