What Liquid Is Denser Than Water
News Leon
Apr 05, 2025 · 5 min read
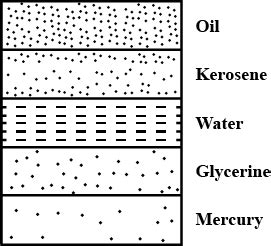
Table of Contents
What Liquid is Denser Than Water? Exploring the World of Density
Density, a fundamental concept in physics and chemistry, describes the mass of a substance per unit volume. It's a crucial property that affects numerous phenomena, from the buoyancy of ships to the layering of liquids in a container. While water serves as a common benchmark (with a density of approximately 1 g/cm³ at 4°C), many liquids boast a higher density. This article delves into the fascinating world of liquids denser than water, exploring their properties, applications, and the science behind their higher densities.
Understanding Density and its Implications
Before diving into specific examples, let's solidify our understanding of density. It's calculated as:
Density = Mass / Volume
This simple formula highlights the relationship between a substance's mass and the space it occupies. A higher density indicates that more mass is packed into a given volume. This has significant implications:
- Buoyancy: Objects denser than water will sink, while those less dense will float. This principle is fundamental to understanding marine life and various engineering applications.
- Layering: When immiscible (non-mixing) liquids of different densities are placed together, they will naturally stratify, with the denser liquid settling at the bottom.
- Mixing: The density difference between liquids influences their mixing behavior. Mixing denser liquids with less dense ones can be challenging and often requires external forces like stirring or agitation.
Liquids Significantly Denser Than Water
Numerous liquids exhibit densities substantially higher than that of water. Here are some notable examples, categorized for clarity:
Inorganic Liquids
-
Mercury (Hg): Famously known for its liquid metallic state at room temperature, mercury boasts a density of approximately 13.6 g/cm³, making it over 13 times denser than water. This extreme density stems from the high atomic weight of mercury atoms and their close packing. Historically used in thermometers and barometers, its toxicity has led to its replacement in many applications.
-
Bromine (Br₂): This reddish-brown liquid halogen has a density around 3.1 g/cm³. Its density is attributable to its relatively high atomic mass and the strong intermolecular forces between its molecules. Bromine is used in various industrial processes, including the production of flame retardants and disinfectants.
-
Sulfuric Acid (H₂SO₄): A highly corrosive strong acid, sulfuric acid has a density of approximately 1.84 g/cm³. The high density is a result of the strong intermolecular hydrogen bonding and the compact structure of the sulfuric acid molecules. It finds widespread use in industrial applications, such as fertilizer production and petroleum refining.
-
Carbon Tetrachloride (CCl₄): Though now largely phased out due to its harmful environmental effects, carbon tetrachloride previously saw use as a solvent and refrigerant. Its density is around 1.59 g/cm³, higher than water due to the presence of heavier chlorine atoms.
Organic Liquids
Many organic liquids, especially those containing heavier halogen atoms or dense aromatic rings, demonstrate densities exceeding that of water.
-
Chloroform (CHCl₃): A colorless, volatile liquid, chloroform possesses a density of about 1.48 g/cm³. The presence of three chlorine atoms significantly increases its density compared to water. Its use as an anesthetic is now largely obsolete due to its toxicity.
-
Dibromoethane (C₂H₄Br₂): This colorless liquid has a density approximately 2.18 g/cm³. The two bromine atoms contribute significantly to its high density. It has been used as a fumigant and in other industrial applications, but its use is declining due to its toxicity.
-
Iodomethane (CH₃I): With a density around 2.28 g/cm³, iodomethane is significantly denser than water. The presence of a heavy iodine atom is the main contributor to this high density. It is employed in certain chemical synthesis reactions.
Metal Alloys in Liquid Form
Several metal alloys, when molten, exhibit densities significantly greater than water. These are often used in specialized industrial processes.
-
Molten Lead: Lead, in its liquid state, possesses a density higher than water, although the precise value varies with temperature. Its high density makes it useful in specific industrial applications.
-
Molten Zinc: Similar to molten lead, liquid zinc also demonstrates a higher density than water, contributing to its usefulness in certain metal casting and galvanizing processes.
Factors Influencing Liquid Density
Several factors determine a liquid's density:
-
Molecular Weight: Heavier molecules generally lead to higher densities, as more mass is packed into the same volume.
-
Molecular Packing: Closely packed molecules result in higher density compared to loosely packed ones. The type of intermolecular forces (e.g., hydrogen bonds, van der Waals forces) influences molecular packing.
-
Temperature: Density generally decreases with increasing temperature. This is because the molecules gain kinetic energy and move farther apart, increasing the volume while the mass remains constant.
-
Pressure: Increased pressure usually leads to higher density as it forces molecules closer together, reducing the volume.
Applications of High-Density Liquids
Liquids denser than water find diverse applications in various fields:
-
Industrial Processes: Many of the liquids mentioned above are crucial in various industrial chemical reactions, serving as solvents, catalysts, or reactants.
-
Medical Applications: While some high-density liquids have toxic properties, certain others, in controlled environments, can find applications in medical imaging techniques.
-
Scientific Research: High-density liquids play a role in various scientific experiments and research, particularly in density gradient centrifugation methods.
-
Engineering: The properties of high-density liquids are exploited in various engineering designs, such as in specific hydraulic systems.
Safety Considerations
Many liquids denser than water, such as mercury, bromine, sulfuric acid, and chloroform, are highly toxic or corrosive. Handling these liquids requires stringent safety precautions, including appropriate personal protective equipment (PPE) like gloves, goggles, and lab coats, and working in well-ventilated areas. Disposal of these liquids must also adhere to strict environmental regulations.
Conclusion
The world of liquids denser than water is diverse and fascinating, encompassing a wide range of substances with unique properties and applications. Understanding density and its relationship to molecular structure and intermolecular forces is key to appreciating the behavior of these liquids and their roles in various scientific and industrial processes. While their high density makes them useful, safety precautions are paramount when handling these potentially hazardous materials. Further research continually expands our knowledge of these liquids and their potential uses. This article serves as an introduction to this intricate field, highlighting some key examples and the science behind their remarkable densities. Continued exploration of these substances promises further advancements and discoveries in various scientific and industrial domains.
Latest Posts
Latest Posts
-
Which Statements Are Correct Check All That Apply
Apr 06, 2025
-
Which Best Describes An Electrical Current
Apr 06, 2025
-
The Largest Endocrine Gland Is The
Apr 06, 2025
-
An Organism Composed Of Only One Cell
Apr 06, 2025
-
Bcc Unit Cell Number Of Atoms
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Liquid Is Denser Than Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
