What Is The Number Of Protons Of Magnesium
News Leon
Apr 05, 2025 · 5 min read
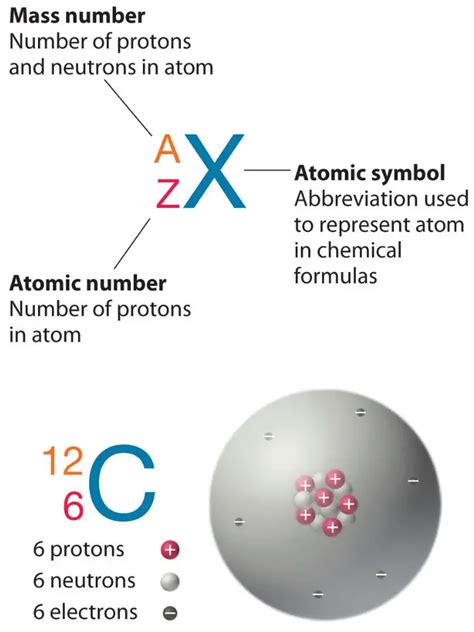
Table of Contents
What is the Number of Protons of Magnesium? A Deep Dive into Atomic Structure
Magnesium, a silvery-white metal essential for life, plays a crucial role in various biological processes and industrial applications. Understanding its atomic structure, particularly the number of protons it possesses, is fundamental to comprehending its chemical behavior and properties. This article delves deep into the atomic structure of magnesium, explaining what defines its atomic number, exploring its isotopes, and discussing its significance in various fields.
Understanding Atomic Number and Protons
The atomic number of an element is arguably its most fundamental property. It represents the number of protons found in the nucleus of a single atom of that element. Protons, along with neutrons, constitute the atom's nucleus, while electrons orbit the nucleus in specific energy levels or shells. The number of protons uniquely identifies an element; no two elements share the same atomic number. Changing the number of protons fundamentally changes the element itself.
For magnesium (Mg), the atomic number is 12. This definitively means that every magnesium atom contains 12 protons in its nucleus. This is a cornerstone of magnesium's chemical identity and dictates its position on the periodic table. Its placement in the periodic table, Group 2 (alkaline earth metals), is directly related to its 2 valence electrons, a consequence of its 12 protons and electron configuration.
Isotopes: Variations in Neutron Count
While the number of protons remains constant for a given element, the number of neutrons can vary. These variations create isotopes. Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. This difference in neutron number affects the atom's mass but not its chemical properties. Magnesium has three naturally occurring stable isotopes:
-
Magnesium-24 (²⁴Mg): This is the most abundant isotope, comprising approximately 79% of naturally occurring magnesium. It contains 12 protons and 12 neutrons (24 - 12 = 12).
-
Magnesium-25 (²⁵Mg): This isotope accounts for about 10% of naturally occurring magnesium. It has 12 protons and 13 neutrons (25 - 12 = 13).
-
Magnesium-26 (²⁶Mg): The least abundant stable isotope, making up roughly 11% of naturally occurring magnesium. It contains 12 protons and 14 neutrons (26 - 12 = 14).
Several radioactive isotopes of magnesium also exist, but these are less common and have shorter half-lives. The isotopic composition of magnesium can vary slightly depending on the source material, but the overall abundance of the three main isotopes remains relatively consistent.
Magnesium's Electron Configuration and Chemical Properties
The 12 protons in a magnesium atom attract 12 electrons, resulting in a specific electron configuration. This configuration determines magnesium's chemical behavior and reactivity. The electron configuration of magnesium is 1s²2s²2p⁶3s². This means that:
- The first energy level (n=1) contains 2 electrons.
- The second energy level (n=2) contains 8 electrons.
- The third energy level (n=3) contains 2 electrons.
The two electrons in the outermost shell (3s²) are the valence electrons. These valence electrons are responsible for magnesium's reactivity. Magnesium readily loses these two valence electrons to achieve a stable octet configuration, similar to the noble gas neon. This tendency to lose electrons makes magnesium a highly reactive metal, readily forming ionic compounds with other elements, particularly non-metals.
Chemical Reactions and Compound Formation
Magnesium's reactivity is evident in its diverse range of chemical reactions. Its ability to lose two electrons leads to the formation of Mg²⁺ ions, which are relatively stable. For instance, magnesium reacts vigorously with oxygen (O₂) in the air to form magnesium oxide (MgO):
2Mg(s) + O₂(g) → 2MgO(s)
Similarly, it reacts with acids like hydrochloric acid (HCl) to produce magnesium chloride (MgCl₂) and hydrogen gas (H₂):
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
These reactions are exothermic, releasing significant amounts of heat. The formation of these ionic compounds highlights the significance of magnesium's 12 protons in determining its chemical properties and reactivity.
The Significance of Magnesium in Biology and Industry
The presence of 12 protons in magnesium's nucleus is not merely an abstract concept; it directly impacts its biological and industrial importance. Magnesium's unique properties stemming from its atomic structure make it vital in various aspects of life and technology.
Biological Roles of Magnesium
Magnesium is an essential mineral for all living organisms. It plays a vital role in hundreds of enzymatic reactions, acting as a cofactor for many enzymes involved in DNA replication, RNA transcription, protein synthesis, and energy metabolism. These crucial roles depend directly on the chemical properties of the magnesium ion (Mg²⁺), a consequence of its 12 protons. Magnesium also plays a critical role in muscle contraction, nerve impulse transmission, and blood sugar control. Deficiency in magnesium can lead to several health problems, emphasizing its crucial importance in biological processes.
Industrial Applications of Magnesium
Magnesium's lightweight nature, high strength-to-weight ratio, and excellent machinability make it a highly sought-after material in various industries. It is widely used in:
-
Automotive Industry: Magnesium alloys are used to produce lightweight car parts, improving fuel efficiency and performance.
-
Aerospace Industry: Its lightweight and high strength properties make it ideal for aircraft and spacecraft components.
-
Electronics Industry: Magnesium is employed in electronic devices due to its electromagnetic shielding properties.
-
Medical Implants: Biocompatible magnesium alloys are increasingly used in biodegradable implants due to their ability to degrade safely within the body.
These applications depend heavily on the material properties of magnesium, directly related to its atomic structure and the presence of 12 protons within its nucleus.
Conclusion: The Defining Role of 12 Protons
The number of protons in an atom defines its elemental identity, and for magnesium, this number is 12. This seemingly simple fact underpins magnesium's chemical behavior, reactivity, and consequently its significance in biology and various industrial applications. Understanding magnesium's atomic structure, particularly the role of its 12 protons, provides a deeper understanding of its unique properties and its importance in diverse fields, from the intricate processes within living organisms to cutting-edge technological advancements. The consistent presence of 12 protons, combined with variations in neutron numbers, gives rise to magnesium’s various isotopic forms, each contributing to the overall abundance and unique properties of this essential element. Therefore, the simple number 12 – the number of protons – holds a profound significance in defining the essential nature of magnesium.
Latest Posts
Latest Posts
-
How Many Orbitals Are There In The Seventh Shell
Apr 05, 2025
-
Which Of The Following Enzyme Digests Protein
Apr 05, 2025
-
Can You See Ribosomes With A Light Microscope
Apr 05, 2025
-
Which One Of The Following Is Not True
Apr 05, 2025
-
What Are The Functions Of The Contractile Vacuole
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Number Of Protons Of Magnesium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
