The Two Most Abundant Components Of Dry Air Are
News Leon
Apr 03, 2025 · 7 min read
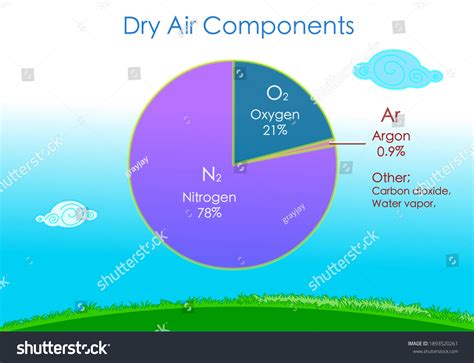
Table of Contents
The Two Most Abundant Components of Dry Air Are… Nitrogen and Oxygen: A Deep Dive
The air we breathe, an invisible yet vital part of our existence, is a complex mixture of gases. While its composition can vary slightly based on location, altitude, and other factors, two components consistently dominate: nitrogen (N₂) and oxygen (O₂). Understanding the properties, roles, and interactions of these gases is crucial to comprehending atmospheric processes, climate change, and the very foundation of life on Earth. This in-depth exploration will delve into the specifics of nitrogen and oxygen, their relative abundances, their importance to life and various industrial processes, and the implications of any changes to their atmospheric concentrations.
Nitrogen (N₂): The Dominant Player
Nitrogen, comprising roughly 78% of dry air by volume, is the undisputed champion in terms of atmospheric abundance. This colorless, odorless, and tasteless gas is a diatomic molecule, meaning two nitrogen atoms are strongly bonded together (N₂). This strong triple bond makes nitrogen remarkably inert under normal conditions, meaning it doesn't readily react with other substances. This inertness is crucial for several reasons:
The Inert Nature of Nitrogen: A Blessing and a Curse
The inertness of nitrogen is both a blessing and a curse. It's a blessing because it prevents rapid oxidation and combustion, safeguarding life and property from uncontrolled reactions. Imagine a world where nitrogen was highly reactive; fires would be far more common and destructive. However, this inertness also presents a challenge: most organisms can't directly utilize atmospheric nitrogen for biological processes.
Nitrogen Fixation: Nature's Essential Conversion
The crucial process of converting atmospheric nitrogen into usable forms is known as nitrogen fixation. This biologically vital transformation is primarily undertaken by specialized bacteria residing in the soil and in the root nodules of certain plants, particularly legumes. These bacteria possess enzymes capable of breaking the strong triple bond in N₂, converting it into ammonia (NH₃) and other nitrogen-containing compounds. This ammonia is then further processed by other microorganisms and plants, ultimately leading to the formation of organic nitrogen compounds crucial for building proteins and nucleic acids—the building blocks of life.
Industrial Applications of Nitrogen
Nitrogen's inertness makes it extremely useful in various industrial applications. It's widely employed:
- As an inert atmosphere: To protect materials susceptible to oxidation, such as food products and electronic components. This prevents spoilage or damage during storage or manufacturing.
- In the production of ammonia: Through the Haber-Bosch process, nitrogen is reacted with hydrogen under high pressure and temperature to produce ammonia (NH₃), a key ingredient in fertilizers.
- In the steel industry: Nitrogen is used to prevent the oxidation of molten steel during manufacturing.
- As a cryogenic refrigerant: Liquid nitrogen, extremely cold at -196°C (-321°F), is utilized in various applications, including cryopreservation and cooling sensitive electronics.
Oxygen (O₂): The Essential Element of Life
Oxygen, making up approximately 21% of dry air by volume, is the second most abundant gas and undeniably crucial for most forms of life on Earth. Unlike nitrogen, oxygen is a highly reactive gas, readily participating in chemical reactions. This reactivity is the key to its vital role in respiration and countless other processes.
Respiration: The Oxygen-Driven Engine of Life
Oxygen's most important role is in aerobic respiration, the process by which most organisms convert nutrients into usable energy. In this process, oxygen acts as the final electron acceptor in a complex series of chemical reactions, liberating energy that powers cellular activities. Without oxygen, this vital energy production process would be severely hampered, resulting in the inability to sustain life as we know it.
The Ozone Layer: Protecting Life from Harmful Radiation
Oxygen also plays a critical role in the formation of the ozone layer (O₃) in the stratosphere. This layer absorbs harmful ultraviolet (UV) radiation from the sun, protecting life on Earth from its damaging effects. The depletion of the ozone layer due to human activities, particularly the release of chlorofluorocarbons (CFCs), is a significant environmental concern with far-reaching consequences for human health and the ecosystem.
Industrial Applications of Oxygen
Oxygen's reactivity and its role as an oxidizing agent are exploited in numerous industrial applications:
- In combustion processes: Oxygen is essential for combustion, driving processes in power generation, metal refining, and various manufacturing procedures.
- In the healthcare industry: Oxygen therapy is crucial in treating respiratory conditions and other medical emergencies.
- In wastewater treatment: Oxygen is used to aid in the decomposition of organic waste.
- In the chemical industry: Oxygen is a reactant in various chemical processes, including the production of numerous compounds.
The Interplay Between Nitrogen and Oxygen: Maintaining Atmospheric Balance
While nitrogen and oxygen are the two most abundant components, the intricate interplay between them, as well as other trace gases, maintains the delicate balance of Earth's atmosphere. This balance is essential for sustaining life and regulating the planet's climate.
Climate Change and Atmospheric Composition
Human activities, especially the burning of fossil fuels, have led to a significant increase in the concentration of greenhouse gases, including carbon dioxide (CO₂), methane (CH₄), and nitrous oxide (N₂O). These gases trap heat in the atmosphere, leading to global warming and climate change. While nitrogen and oxygen are not direct contributors to the enhanced greenhouse effect, changes in their relative concentrations could have indirect effects on atmospheric processes and climate.
Air Pollution and its Impact
Air pollution, caused by the release of various pollutants into the atmosphere, impacts the balance of gases. Certain pollutants react with nitrogen and oxygen, leading to the formation of smog, acid rain, and other harmful substances. This pollution not only affects air quality, but it also has severe consequences for human health and the environment.
Future Considerations: Maintaining Atmospheric Integrity
Maintaining the integrity of Earth's atmosphere, including the balance of nitrogen and oxygen, is crucial for the well-being of both humanity and the planet. This requires a concerted effort to reduce greenhouse gas emissions, mitigate air pollution, and promote sustainable practices. Understanding the properties and roles of nitrogen and oxygen, the most abundant components of dry air, is paramount in addressing these environmental challenges and ensuring the long-term health of our planet.
Other Trace Gases and Their Significance
Beyond nitrogen and oxygen, the atmosphere contains a variety of other gases in much smaller concentrations. These gases, though present in smaller amounts, often play crucial roles in atmospheric chemistry, climate regulation, and various biological processes:
Argon (Ar): The Inert Noble Gas
Argon, making up about 0.93% of dry air, is an inert noble gas that plays relatively little role in biological processes. However, its inertness makes it valuable in various industrial applications, similar to nitrogen.
Carbon Dioxide (CO₂): A Crucial Greenhouse Gas
Carbon dioxide, currently around 0.04% of the atmosphere, is a crucial greenhouse gas. It plays an essential role in regulating the Earth's temperature, but its increasing concentration due to human activities is a primary driver of climate change.
Water Vapor (H₂O): Variable but Important
The concentration of water vapor in the atmosphere is highly variable, depending on location and weather conditions. It’s a potent greenhouse gas and plays a critical role in the water cycle.
Neon (Ne), Helium (He), Methane (CH₄), Krypton (Kr), Hydrogen (H₂), and Xenon (Xe): Trace Gases with Unique Roles
These gases are present in minute quantities but still contribute to the overall composition and behavior of the atmosphere. Helium, for instance, is used in various applications due to its inertness and low density. Methane, a potent greenhouse gas, is released from various sources, including agriculture and natural gas production.
Conclusion: Understanding the Air We Breathe
The composition of air, dominated by nitrogen and oxygen, is not static. Variations in the concentrations of these gases, as well as other trace components, can significantly impact atmospheric processes, climate, and life on Earth. A deeper understanding of these gases, their properties, their interactions, and the implications of changes in their concentrations is vital for addressing current and future environmental challenges. By continuing research and implementing sustainable practices, we can strive to maintain the delicate balance of our atmosphere and ensure a healthy planet for generations to come. The seemingly simple act of breathing relies on a complex and finely tuned atmospheric system, and appreciating its intricacies is a fundamental step towards responsible stewardship of our planet.
Latest Posts
Related Post
Thank you for visiting our website which covers about The Two Most Abundant Components Of Dry Air Are . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
