The Longer The Wavelength The Higher The Energy
News Leon
Apr 02, 2025 · 5 min read
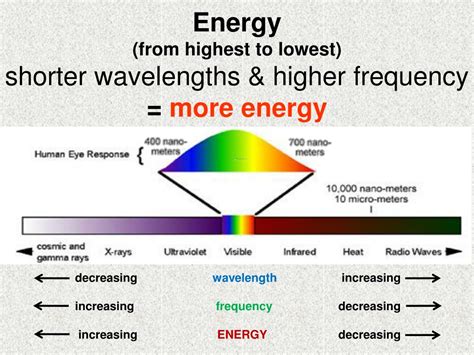
Table of Contents
The Longer the Wavelength, the Higher the Energy? A Deep Dive into the Misconception
The statement "the longer the wavelength, the higher the energy" is incorrect. In fact, the opposite is true for most forms of electromagnetic radiation: the shorter the wavelength, the higher the energy. This common misconception highlights the often counter-intuitive nature of wave-particle duality and the relationship between wavelength, frequency, and energy. This article will delve into the fundamental physics behind this relationship, explore the exceptions to the rule, and clarify the widespread misunderstanding.
Understanding Electromagnetic Radiation
Electromagnetic radiation (EMR) encompasses a broad spectrum of energy, ranging from radio waves with extremely long wavelengths to gamma rays with incredibly short wavelengths. All forms of EMR share the same fundamental properties: they are transverse waves that propagate through space at the speed of light (approximately 3 x 10<sup>8</sup> m/s in a vacuum). These waves consist of oscillating electric and magnetic fields perpendicular to each other and to the direction of propagation.
The Key Relationship: Wavelength, Frequency, and Energy
The key to understanding the energy of EMR lies in the relationship between its wavelength (λ), frequency (ν), and energy (E). These three properties are interconnected by the following fundamental equations:
-
c = λν where 'c' represents the speed of light. This equation states that the speed of light is the product of wavelength and frequency. A longer wavelength means a lower frequency, and vice versa.
-
E = hν where 'h' is Planck's constant (approximately 6.626 x 10<sup>-34</sup> Js). This equation, derived from quantum mechanics, demonstrates that the energy of a photon (a quantum of light) is directly proportional to its frequency. Higher frequency means higher energy.
Combining these two equations, we can express the energy of a photon in terms of its wavelength:
- E = hc/λ
This equation clearly shows the inverse relationship between energy and wavelength. As the wavelength (λ) increases, the energy (E) decreases, and vice versa. A shorter wavelength means a higher frequency, leading to a higher energy photon.
Visualizing the Electromagnetic Spectrum
The electromagnetic spectrum illustrates this relationship beautifully. It's arranged from longest wavelength (lowest energy) to shortest wavelength (highest energy):
- Radio Waves: Longest wavelengths, lowest energy. Used in communication, broadcasting, and medical imaging (MRI).
- Microwaves: Shorter wavelengths than radio waves, higher energy. Used in cooking, communication, and radar.
- Infrared Radiation: Shorter wavelengths than microwaves, higher energy. Experienced as heat, used in thermal imaging and remote controls.
- Visible Light: A narrow band of wavelengths we can see, with violet having the shortest wavelength (highest energy) and red having the longest wavelength (lowest energy).
- Ultraviolet Radiation: Shorter wavelengths than visible light, higher energy. Can cause sunburn and DNA damage.
- X-rays: Much shorter wavelengths than UV, very high energy. Used in medical imaging and material analysis.
- Gamma Rays: Shortest wavelengths, highest energy. Highly penetrating and damaging, emitted by radioactive materials and nuclear reactions.
Why the Misconception?
The confusion might arise from several sources:
- Intuitive Misinterpretation: Our everyday experiences don't often deal with the subtle nuances of quantum mechanics. We tend to associate "bigger" with "stronger" or "more powerful," leading to the incorrect association of longer wavelengths with higher energy.
- Confusing Wave Properties: Wavelength and amplitude are often conflated. While wavelength determines energy, amplitude determines intensity (brightness or loudness). A wave with a large amplitude carries more intensity but not necessarily more energy per photon.
- Lack of Clear Visual Representation: While the electromagnetic spectrum visually demonstrates the wavelength-energy relationship, the subtle differences in wavelengths might not be immediately apparent to everyone.
Exceptions and Nuances
While the inverse relationship between wavelength and energy holds true for most EMR, some nuances and exceptions exist:
- Gravitational Waves: These waves, predicted by Einstein's theory of general relativity, are not electromagnetic in nature. Their energy relationship with wavelength is different and more complex.
- Matter Waves: According to de Broglie's hypothesis, matter also exhibits wave-like behavior. The wavelength of a matter wave is inversely proportional to its momentum, not directly related to its energy in the same way as electromagnetic waves.
Practical Applications and Importance
Understanding the relationship between wavelength and energy is crucial in numerous fields:
- Medicine: Different types of EMR are used for diagnosis and treatment, each chosen based on its energy and penetrating power. X-rays, for example, are used for imaging bones due to their high energy and ability to penetrate soft tissue.
- Astronomy: Analyzing the wavelengths of light from stars and galaxies helps determine their temperature, composition, and motion. The observation of specific wavelengths allows scientists to identify elements present in celestial bodies.
- Communications: Different wavelengths are used for different communication technologies, from radio waves for long-distance broadcasting to microwaves for satellite communication. The choice depends on the desired range and data transmission speed.
- Material Science: The interaction of EMR with materials depends on the wavelength of the radiation and the properties of the material. This knowledge is essential in designing new materials and technologies.
Conclusion: Clearing the Confusion
The statement "the longer the wavelength, the higher the energy" is a fundamental misconception regarding electromagnetic radiation. The accurate statement is: the shorter the wavelength, the higher the energy. This inverse relationship stems from the fundamental equations connecting wavelength, frequency, and energy. While some exceptions exist, the general principle holds true across a vast range of applications and scientific fields. Understanding this relationship is crucial for comprehending the behavior of light and its interactions with matter, ultimately underpinning many advancements in science and technology. By clarifying this common misconception, we hope to provide a clearer and more accurate understanding of the fascinating world of electromagnetic radiation. Further exploration into quantum mechanics and wave-particle duality can further enhance this knowledge and dispel lingering ambiguities.
Latest Posts
Latest Posts
-
A Solution With A Ph Of 5 0 Is
Apr 03, 2025
-
Earth Is Inclined At An Angle Of
Apr 03, 2025
-
Which Form Of Energy Increases When A Spring Is Compressed
Apr 03, 2025
-
Which Of The Following Best Describes The Structure Of Dna
Apr 03, 2025
-
Declarative Imperative Interrogative And Exclamatory Sentences
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Longer The Wavelength The Higher The Energy . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
