Red Blood Cells Placed In A Hypotonic Solution Will
News Leon
Apr 04, 2025 · 5 min read
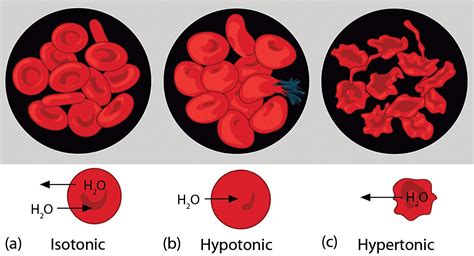
Table of Contents
Red Blood Cells Placed in a Hypotonic Solution Will… Undergo Hemolysis!
Understanding the behavior of red blood cells (RBCs, also known as erythrocytes) in different solutions is crucial for comprehending fundamental concepts in biology and medicine. This article delves deep into the effects of placing red blood cells in a hypotonic solution, explaining the underlying mechanisms, the observable changes, and the broader implications of this phenomenon.
What is a Hypotonic Solution?
Before exploring the effects on RBCs, let's define our terms. A hypotonic solution is one that has a lower concentration of solutes (dissolved substances) compared to another solution, separated by a selectively permeable membrane, such as the cell membrane. Think of it as a solution with a relatively higher concentration of water. This difference in solute concentration creates an osmotic gradient, driving the movement of water across the membrane.
Osmosis: The Driving Force
Osmosis is the passive movement of water across a selectively permeable membrane from a region of high water concentration (low solute concentration) to a region of low water concentration (high solute concentration). This movement continues until equilibrium is reached, meaning the concentration of water is equal on both sides of the membrane. The driving force behind osmosis is the difference in water potential between the two solutions.
Red Blood Cells: A Brief Overview
Red blood cells are specialized cells designed for oxygen transport. Their unique biconcave disc shape maximizes surface area for efficient gas exchange. The cell membrane of a RBC is selectively permeable, allowing certain substances to pass through while restricting others. This membrane plays a critical role in maintaining the cell's integrity and function.
Placing RBCs in a Hypotonic Solution: The Hemolysis Process
When red blood cells are placed in a hypotonic solution, the concentration of water outside the cell is higher than inside. This creates an osmotic gradient, causing water to move across the cell membrane into the RBC. The influx of water causes the cell to swell.
Stages of Hemolysis:
-
Initial Swelling: The cell initially swells as water enters, increasing its volume. The cell membrane is flexible and can accommodate some expansion.
-
Increased Turgor Pressure: The increasing internal pressure, known as turgor pressure, pushes against the cell membrane.
-
Membrane Rupture (Hemolysis): If the influx of water continues, the cell membrane eventually reaches its elastic limit and ruptures. This process is called hemolysis, and the contents of the cell, including hemoglobin, are released into the surrounding solution. The solution becomes visibly reddish.
-
Ghost Cells: After hemolysis, the remaining cell membrane forms a "ghost cell," a pale, empty shell of the original RBC.
Visualizing the Process
Imagine a balloon (representing the RBC) partially filled with water (representing the intracellular fluid). If you place this balloon in a large container filled with more water, the balloon will expand as water enters. If enough water enters, the balloon will eventually burst, similar to the hemolysis of an RBC in a hypotonic solution.
Factors Affecting Hemolysis Rate
The rate at which hemolysis occurs depends on several factors:
-
Degree of Hypotonicity: The greater the difference in solute concentration between the solution and the inside of the RBC, the faster the water influx and the quicker the hemolysis.
-
Temperature: Higher temperatures generally accelerate the rate of osmosis, leading to faster hemolysis.
-
Cell Age: Older RBCs are more susceptible to hemolysis than younger, more robust cells. Their membranes may be less flexible and more prone to rupture.
-
Presence of other substances: Certain substances in the solution can influence membrane permeability and affect the hemolysis rate.
Importance in Biological and Medical Contexts
The understanding of hemolysis has significant implications in various fields:
-
Blood Transfusions: Incorrectly matching blood types can lead to hemolysis as antibodies in the recipient's plasma react with antigens on the donor's RBCs, causing cell rupture.
-
Clinical Diagnosis: Hemolysis can be an indicator of various medical conditions, including hemolytic anemia (a group of disorders characterized by the premature destruction of red blood cells), and certain infections. Measuring the degree of hemolysis can be used in diagnostic tests.
-
Drug Development: Research on the effects of drugs and other substances on RBC membranes involves studies of hemolysis to assess their toxicity and potential side effects.
-
Cell Biology Research: Hemolysis is a crucial technique in cell biology experiments. It allows researchers to isolate and study various components of RBCs, such as hemoglobin and membrane proteins.
-
Water Balance Regulation: The understanding of osmosis and hemolysis is vital to understanding how organisms maintain water balance. The ability of cells to withstand changes in osmotic pressure is a critical aspect of survival.
Preventing Hemolysis
In situations where hemolysis is undesirable, such as during blood transfusions or in cell culture experiments, several strategies can be employed to prevent it:
-
Isotonic Solutions: Using an isotonic solution, which has the same solute concentration as the inside of the RBC, prevents net water movement and thus prevents hemolysis or crenation (shrinking of cells). Normal saline (0.9% NaCl) is a common isotonic solution used in medical settings.
-
Careful Handling of Blood Samples: Gentle handling and proper storage of blood samples minimizes mechanical damage to RBCs, reducing the risk of hemolysis.
-
Controlled Environmental Conditions: Maintaining appropriate temperature and avoiding exposure to harsh chemicals can minimize hemolysis during experiments or storage.
Conclusion
The behavior of red blood cells in a hypotonic solution, resulting in hemolysis, provides a clear demonstration of the principles of osmosis and the importance of maintaining osmotic balance in biological systems. The process of hemolysis and its consequences have profound implications in various areas of biology and medicine, highlighting the critical role of red blood cells in health and disease. Understanding this fundamental process allows for advances in medical treatments, diagnostic techniques, and overall biological knowledge. Further research continues to explore the intricacies of this fascinating phenomenon and its various applications. From blood transfusions to disease diagnostics, the implications of hemolysis are far-reaching and continue to shape our understanding of cellular processes and their impact on human health.
Latest Posts
Latest Posts
-
Which Of The Following Is True For Displacement
Apr 05, 2025
-
Which Of The Following Is An Example Of A Browser
Apr 05, 2025
-
Essay On The Pleasure Of Reading
Apr 05, 2025
-
What Three Elements Make Up Carbohydrates
Apr 05, 2025
-
The Movement Of People From One Place To Another
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Red Blood Cells Placed In A Hypotonic Solution Will . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
