Positively Charged Center Of An Atom
News Leon
Apr 03, 2025 · 6 min read
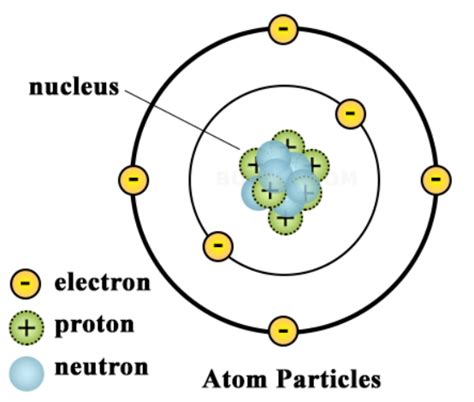
Table of Contents
Positively Charged Center of an Atom: Delving into the Nucleus
The atom, the fundamental building block of matter, is a fascinating microcosm of complex interactions. At its heart lies the nucleus, a tiny but incredibly dense region brimming with positive charge. Understanding this positively charged center is crucial to grasping the nature of matter, chemical reactions, and the very fabric of the universe. This article will delve deep into the nucleus, exploring its composition, properties, and significance in various scientific fields.
Unveiling the Nucleus: Composition and Structure
The positively charged center of an atom, the nucleus, is composed of two primary types of particles: protons and neutrons. These particles, collectively known as nucleons, are bound together by a powerful force known as the strong nuclear force. This force is significantly stronger than the electromagnetic force, which would otherwise cause the positively charged protons to repel each other and the nucleus to fly apart.
Protons: The Positive Charge Carriers
Protons are subatomic particles carrying a single positive charge, equal in magnitude but opposite in sign to the charge of an electron. Each proton contributes significantly to the overall positive charge of the nucleus. The number of protons in an atom's nucleus defines its atomic number, a fundamental property that determines the element's identity. For instance, an atom with one proton is hydrogen, an atom with six protons is carbon, and an atom with 92 protons is uranium. The proton's mass is approximately 1 atomic mass unit (amu).
Neutrons: The Neutral Stabilizers
Neutrons, as their name suggests, carry no net electric charge. They are slightly more massive than protons, with a mass of approximately 1 amu. Neutrons play a crucial role in nuclear stability. The strong nuclear force, which binds protons and neutrons together, is most effective when there's a balanced or near-balanced number of protons and neutrons. Isotopes, variations of an element with different numbers of neutrons, showcase this influence. While some isotopes are stable, others are radioactive due to an imbalance in the proton-neutron ratio, leading to nuclear decay.
The Strong Nuclear Force: The Glue that Holds the Nucleus Together
The strong nuclear force is a fundamental force of nature responsible for binding protons and neutrons within the nucleus. It's a short-range force, meaning its strength diminishes rapidly with increasing distance between nucleons. This characteristic is crucial for the stability of the nucleus. If the strong nuclear force were long-range, it would bind all protons and neutrons in the universe together.
Overcoming Electromagnetic Repulsion
The strong nuclear force is essential because it overcomes the electromagnetic repulsion between positively charged protons. Protons, possessing the same charge, naturally repel each other according to Coulomb's law. The strong nuclear force is much stronger than the electromagnetic force at short distances within the nucleus, effectively countering the repulsive forces and keeping the nucleus intact.
Nuclear Binding Energy: A Measure of Stability
The energy required to disassemble a nucleus into its constituent protons and neutrons is known as nuclear binding energy. This energy represents the strength of the strong nuclear force holding the nucleus together. Nuclei with high binding energy per nucleon are more stable than those with low binding energy per nucleon. This concept explains the stability and radioactivity of various isotopes.
Exploring Nuclear Properties: Size, Density, and Stability
The nucleus occupies a remarkably small fraction of the atom's total volume. Despite its diminutive size, it contains almost all of the atom's mass. The density of the nucleus is incredibly high, on the order of 10¹⁷ kg/m³, far surpassing the density of any known material.
Isotopes and Radioactive Decay
Isotopes, as mentioned earlier, are atoms of the same element with the same number of protons but different numbers of neutrons. Some isotopes are stable, while others are unstable or radioactive. Radioactive isotopes undergo radioactive decay, emitting particles or energy to transform into a more stable configuration. Types of radioactive decay include alpha decay, beta decay, and gamma decay. These decay processes are governed by the weak nuclear force.
Nuclear Fission and Fusion: Harnessing Nuclear Energy
The immense energy stored within the nucleus can be harnessed through nuclear reactions. Nuclear fission involves splitting a heavy nucleus into two smaller nuclei, releasing a large amount of energy. This process is utilized in nuclear power plants and atomic bombs. Nuclear fusion, on the other hand, involves combining two light nuclei to form a heavier nucleus, also releasing a substantial amount of energy. This process powers the sun and other stars.
The Nucleus and its Significance in Science and Technology
The nucleus plays a pivotal role in various scientific and technological fields. Its properties are fundamental to understanding:
- Chemistry: The number of protons determines an element's chemical properties and its behavior in chemical reactions. Isotopes can have slightly different chemical properties due to their mass differences, leading to applications like isotopic tracing.
- Nuclear Physics: Nuclear physics explores the structure and behavior of atomic nuclei, aiming to understand the strong and weak nuclear forces and the processes of nuclear reactions.
- Nuclear Medicine: Radioactive isotopes are used in medical imaging and therapy. Techniques like PET (positron emission tomography) and radiotherapy rely on the properties of radioactive nuclei.
- Nuclear Energy: Nuclear fission is used to generate electricity in nuclear power plants, providing a significant source of energy worldwide. Research into nuclear fusion aims to develop a sustainable and clean energy source.
- Geology and Archaeology: Radioactive decay provides a powerful tool for dating rocks and artifacts, allowing scientists to determine the age of geological formations and ancient objects.
Beyond Protons and Neutrons: Exploring Subatomic Particles
While protons and neutrons were once considered fundamental particles, further research revealed that they are composed of even smaller particles called quarks. Protons and neutrons each consist of three quarks: two up quarks and one down quark for a proton, and one up quark and two down quarks for a neutron. The interaction of quarks is mediated by gluons, the force carriers of the strong nuclear force.
The Standard Model of Particle Physics
The Standard Model of particle physics provides a comprehensive framework for understanding the fundamental constituents of matter and their interactions. It incorporates quarks, leptons (such as electrons), and the force-carrying bosons (such as photons, gluons, and W and Z bosons). The model has been remarkably successful in explaining experimental results, but it also leaves some questions unanswered, such as the nature of dark matter and dark energy.
Conclusion: The Nucleus – A Microcosm of the Universe
The positively charged center of an atom, the nucleus, is a fascinating and complex structure that holds the key to understanding the behavior of matter. Its composition, properties, and the forces governing it are essential to numerous scientific fields. From nuclear energy to medical applications to our understanding of the universe itself, the study of the nucleus continues to reveal insights into the fundamental laws of nature. Further research will undoubtedly unveil deeper secrets of this tiny but incredibly powerful component of the atom. Further study into the strong and weak nuclear forces, the behavior of quarks and gluons, and the ongoing search for a unified theory encompassing all fundamental forces will continue to shape our understanding of the nucleus and its profound impact on the universe. The nucleus, once a simple concept, now stands as a testament to the intricate and beautiful complexity of the natural world. Its continued investigation remains a cornerstone of scientific progress, promising exciting discoveries and technological advancements in the years to come. The journey to fully understanding the nucleus is far from over, and each new discovery offers a deeper appreciation for the fundamental forces that govern our universe.
Latest Posts
Latest Posts
-
Which Of The Following Is Not True Of Dna
Apr 04, 2025
-
Any Computer Parts That You Can Actually Touch
Apr 04, 2025
-
Which Of The Following Is Hydrophobic
Apr 04, 2025
-
Do Prokaryotes Have A Membrane Bound Organelles
Apr 04, 2025
-
Why Was A Stain Added To The Cheek Cells
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Positively Charged Center Of An Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
