Is Sugar A Compound Mixture Or Element
News Leon
Apr 02, 2025 · 5 min read
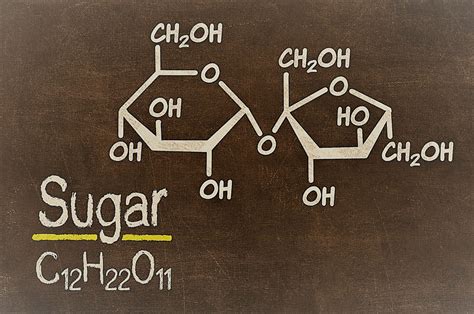
Table of Contents
- Is Sugar A Compound Mixture Or Element
- Table of Contents
- Is Sugar a Compound, Mixture, or Element? A Deep Dive into Sucrose
- Understanding the Building Blocks of Matter: Elements, Compounds, and Mixtures
- Elements
- Compounds
- Mixtures
- The Chemical Composition of Sugar (Sucrose)
- The Covalent Bonds in Sucrose
- Why Sugar is NOT a Mixture or Element
- Different Types of Sugars: Still Compounds
- The Importance of Understanding Sugar's Chemical Nature
- Conclusion: Sugar – A Compound with Diverse Applications
- Latest Posts
- Latest Posts
- Related Post
Is Sugar a Compound, Mixture, or Element? A Deep Dive into Sucrose
Sugar, a ubiquitous substance in our diets and countless industrial processes, often sparks curiosity about its fundamental nature. Is it an element, a compound, or a mixture? Understanding this requires exploring the basic building blocks of matter and the properties that define each category. This comprehensive guide will delve into the chemical composition of sugar, specifically sucrose, to definitively answer this question and explore related concepts.
Understanding the Building Blocks of Matter: Elements, Compounds, and Mixtures
Before classifying sugar, let's establish a clear understanding of the three fundamental categories of matter:
Elements
Elements are pure substances composed of only one type of atom. Atoms are the smallest units of matter that retain the chemical properties of an element. They are listed on the periodic table, with each element possessing a unique atomic number representing the number of protons in its nucleus. Examples include oxygen (O), carbon (C), and hydrogen (H). Elements cannot be broken down into simpler substances through chemical means.
Compounds
Compounds are pure substances formed when two or more elements chemically combine in a fixed ratio. This combination involves the sharing or transfer of electrons between atoms, creating strong chemical bonds. These bonds determine the properties of the compound, which are often vastly different from the properties of the constituent elements. Compounds can be broken down into simpler substances through chemical reactions, such as electrolysis or combustion. Water (H₂O), for example, is a compound composed of hydrogen and oxygen atoms in a 2:1 ratio.
Mixtures
Mixtures are physical combinations of two or more substances that are not chemically bonded. The components retain their individual properties, and the proportions can vary. Mixtures can be homogeneous (uniform throughout, like saltwater) or heterogeneous (non-uniform, like sand and water). Mixtures can be separated into their components through physical methods like filtration, distillation, or evaporation.
The Chemical Composition of Sugar (Sucrose)
The common table sugar we use is primarily sucrose, a compound. Its chemical formula is C₁₂H₂₂O₁₁. This formula reveals that each molecule of sucrose is composed of:
- 12 carbon atoms (C)
- 22 hydrogen atoms (H)
- 11 oxygen atoms (O)
These atoms are not simply mixed together; they are chemically bonded in a specific arrangement to form a stable molecule. This arrangement gives sucrose its unique properties, including its sweetness, solubility in water, and its ability to undergo various chemical reactions.
The Covalent Bonds in Sucrose
The atoms within a sucrose molecule are held together by covalent bonds. Covalent bonds involve the sharing of electrons between atoms. In sucrose, carbon, hydrogen, and oxygen atoms share electrons to achieve a stable electron configuration. This sharing creates a strong, stable structure characteristic of compounds. The specific arrangement of these bonds contributes to sucrose's crystalline structure and its other properties.
Why Sugar is NOT a Mixture or Element
Let's address why sugar isn't a mixture or an element:
-
Not a Mixture: Sugar is not a mixture because its components (carbon, hydrogen, and oxygen) are not simply mixed together physically. They are chemically bonded in a fixed ratio to form a distinct molecule with unique properties. You cannot physically separate sucrose into its constituent elements without breaking chemical bonds.
-
Not an Element: Sugar is not an element because it's composed of more than one type of atom. Elements contain only one type of atom, whereas sucrose contains three different types: carbon, hydrogen, and oxygen.
Different Types of Sugars: Still Compounds
While sucrose is the most common table sugar, other sugars exist, each with its own chemical formula and properties. These include:
-
Glucose (C₆H₁₂O₆): A simple sugar, also known as dextrose, found in fruits and honey. It's a crucial source of energy for living organisms.
-
Fructose (C₆H₁₂O₆): Another simple sugar, known as fruit sugar, found naturally in fruits and honey. It's sweeter than glucose.
-
Lactose (C₁₂H₂₂O₁₁): A disaccharide (a sugar composed of two simpler sugars) found in milk. It's composed of glucose and galactose.
All these sugars are compounds, each composed of different elements chemically bonded together in specific ratios. They share similarities (they are all carbohydrates, containing carbon, hydrogen, and oxygen) but possess unique chemical structures and properties.
The Importance of Understanding Sugar's Chemical Nature
Understanding that sugar is a compound, not a mixture or element, has several significant implications:
-
Nutritional Science: Knowing the chemical composition of sugars helps us understand their role in metabolism and energy production in the body. The different types of sugars are processed differently by the body, affecting blood sugar levels and overall health.
-
Food Science and Technology: Understanding sugar's chemical properties allows food scientists and technologists to manipulate its behavior in food processing, influencing texture, sweetness, and preservation.
-
Industrial Applications: Sucrose's chemical properties enable its use in a vast range of industrial applications, including the production of ethanol, pharmaceuticals, and textiles.
Conclusion: Sugar – A Compound with Diverse Applications
In conclusion, sugar, specifically sucrose, is unequivocally a compound. It's a pure substance formed by the chemical combination of carbon, hydrogen, and oxygen atoms in a fixed ratio (C₁₂H₂₂O₁₁). Its chemical structure determines its unique properties and its diverse applications in various fields. Understanding the fundamental differences between elements, compounds, and mixtures is crucial for comprehending the nature of matter and the behavior of substances like sugar. The chemical composition of sugar, its bonding characteristics, and its role in biological and industrial processes highlight the significance of chemistry in our daily lives. Further exploration of carbohydrate chemistry and related fields will reveal even more intriguing aspects of this seemingly simple molecule.
Latest Posts
Latest Posts
-
What Is Found In Both Eukaryotic And Prokaryotic Cells
Apr 04, 2025
-
Select The Correct Electron Configuration For Cr
Apr 04, 2025
-
Can A Pure Substance Be A Compound
Apr 04, 2025
-
3 Types Of Winds Class 7
Apr 04, 2025
-
The Tendency Of Chemicals To Become More Concentrated
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Is Sugar A Compound Mixture Or Element . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
