In A Hypotonic Solution An Animal Cell Will
News Leon
Apr 03, 2025 · 5 min read
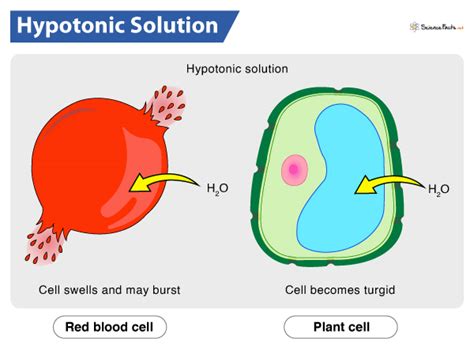
Table of Contents
In a Hypotonic Solution, an Animal Cell Will… Lyse! Understanding Osmosis and Cell Behavior
Understanding how cells react to different environments is crucial in biology. One key concept is osmosis, the movement of water across a semipermeable membrane from an area of high water concentration to an area of low water concentration. This movement is driven by the difference in water potential, aiming to achieve equilibrium. This article delves into the effects of a hypotonic solution on animal cells, explaining the process, the consequences, and the broader implications for cell biology and physiology.
What is a Hypotonic Solution?
A hypotonic solution is one where the concentration of solutes (dissolved substances) is lower outside the cell than inside the cell. This means the water concentration is higher outside the cell. Think of it like this: the solution has a lower solute potential and a higher water potential compared to the cell's internal environment. This difference in water potential is the driving force behind the movement of water.
Understanding Water Potential
Water potential is a measure of the tendency of water to move from one area to another. It's influenced by two main factors:
-
Solute potential: This reflects the effect of dissolved solutes on water potential. The more solutes present, the lower the solute potential (more negative).
-
Pressure potential: This refers to the pressure exerted on water. Positive pressure potential (turgor pressure) pushes water out, while negative pressure potential (tension) pulls water in.
In a hypotonic solution, the water potential outside the cell is higher (less negative) than inside the cell. This difference creates an osmotic gradient, driving water into the cell.
Osmosis in Animal Cells: The Hypotonic Effect
When an animal cell is placed in a hypotonic solution, water rushes into the cell via osmosis. This influx of water causes the cell to swell. Animal cells lack a rigid cell wall, unlike plant cells, which provides structural support. This lack of a rigid outer layer makes animal cells particularly vulnerable to the effects of a hypotonic environment.
The Consequences: Lysis
The continuous influx of water in a hypotonic solution leads to lysis, the bursting of the cell. As the cell swells, its plasma membrane stretches, eventually reaching its breaking point. The cell's contents spill out into the surrounding solution, resulting in cell death. This process is dramatic and irreversible, highlighting the sensitivity of animal cells to osmotic imbalances.
Contrasting with Other Solutions: Isotonic and Hypertonic
To fully appreciate the effects of a hypotonic solution, it’s important to compare it with isotonic and hypertonic solutions:
-
Isotonic solution: In an isotonic solution, the solute concentration is equal inside and outside the cell. There's no net movement of water, and the cell maintains its shape and size. This is the ideal environment for many animal cells.
-
Hypertonic solution: In a hypertonic solution, the solute concentration is higher outside the cell than inside. Water moves out of the cell via osmosis, causing the cell to shrink and potentially crenate (develop a shriveled appearance). This process is also detrimental to cell function and can lead to cell death.
Mechanisms Protecting Against Lysis: A Closer Look
While lysis is a significant threat in hypotonic environments, certain mechanisms help some cells to mitigate the effects of osmotic stress:
-
Ion channels and pumps: Cells actively regulate the movement of ions across their membranes using ion channels and pumps. This helps to control the internal osmotic pressure and prevent excessive water influx. These mechanisms are crucial for maintaining osmotic balance.
-
Aquaporins: These specialized water channels facilitate the rapid movement of water across the cell membrane. While primarily involved in water uptake, their regulation can help control the rate of water influx in a hypotonic environment. A finely tuned system of aquaporin activity helps to prevent lysis.
Real-World Examples and Applications
The understanding of osmosis and hypotonic solutions has wide-ranging applications across various fields:
-
Medicine: Intravenous fluids are carefully formulated to be isotonic to avoid damaging red blood cells. A hypotonic solution administered intravenously could lead to the lysis of red blood cells, causing serious complications.
-
Agriculture: Understanding the osmotic effects of different soil solutions is vital for optimizing crop growth and yield. Farmers need to maintain appropriate soil salinity to prevent osmotic stress on plant roots.
-
Food preservation: Osmosis is used in preserving food items such as fruits and vegetables. The application of a hypertonic solution draws water out of the food, inhibiting microbial growth and increasing shelf life.
Beyond Lysis: Sublethal Effects of Hypotonic Solutions
While lysis is the most severe outcome of exposing animal cells to a hypotonic solution, sublethal effects can also occur. These effects might not immediately result in cell death but can still significantly impair cell function:
-
Cell swelling and deformation: Even before lysis, significant cell swelling can disrupt intracellular structures and interfere with normal cellular processes.
-
Disruption of membrane function: The stretching of the plasma membrane due to water influx can compromise its integrity and function, affecting transport processes and signaling pathways.
-
Cytoskeletal disruption: The cell's cytoskeleton, a network of protein filaments providing structural support, can be disrupted by excessive cell swelling, leading to loss of cell shape and impaired motility.
Research and Future Directions
Ongoing research continues to explore the intricate details of cellular responses to osmotic stress. Areas of investigation include:
-
The role of specific ion channels and transporters in regulating osmotic balance. Understanding the precise mechanisms involved could lead to new therapeutic strategies for treating diseases related to osmotic imbalances.
-
The development of novel methods for protecting cells from osmotic stress. This could have significant applications in various fields, including medicine and biotechnology.
-
The impact of osmotic stress on cell signaling pathways. Understanding how osmotic stress affects cell signaling could provide insights into the underlying mechanisms of various diseases.
Conclusion: A Delicate Balance
The response of an animal cell to a hypotonic solution underscores the importance of maintaining a delicate osmotic balance. While the process of osmosis is essential for cellular function, uncontrolled water influx can have catastrophic consequences, leading to lysis and cell death. A comprehensive understanding of osmosis, the factors influencing water potential, and the various cellular mechanisms involved is crucial in various fields, from medicine to agriculture. Further research promises to unravel even more of the intricacies of this fundamental biological process and its implications for cell health and survival.
Latest Posts
Latest Posts
-
Which Of The Following Is Not True Of Dna
Apr 04, 2025
-
Any Computer Parts That You Can Actually Touch
Apr 04, 2025
-
Which Of The Following Is Hydrophobic
Apr 04, 2025
-
Do Prokaryotes Have A Membrane Bound Organelles
Apr 04, 2025
-
Why Was A Stain Added To The Cheek Cells
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about In A Hypotonic Solution An Animal Cell Will . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
