How Many Valence Electrons Does Oxygen
News Leon
Apr 05, 2025 · 5 min read
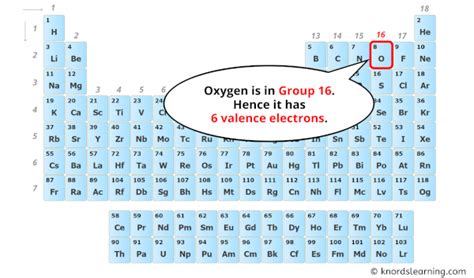
Table of Contents
How Many Valence Electrons Does Oxygen Have? A Deep Dive into Atomic Structure
Oxygen, a life-sustaining element crucial for respiration and countless biological processes, holds a fascinating position in the periodic table. Understanding its atomic structure, specifically the number of valence electrons, is key to grasping its chemical behavior and reactivity. This article delves deep into the question: How many valence electrons does oxygen have? We'll explore the concept of valence electrons, the electron configuration of oxygen, and how this determines its bonding properties and role in various chemical reactions.
Understanding Valence Electrons: The Key to Chemical Bonding
Before we pinpoint the number of valence electrons in oxygen, let's clarify what valence electrons are. Valence electrons are the electrons located in the outermost shell, or energy level, of an atom. These electrons are the most loosely bound and, therefore, are the ones primarily involved in chemical bonding. They dictate how an atom will interact with other atoms, determining its reactivity and the types of chemical bonds it can form. The number of valence electrons directly influences an element's chemical properties. Atoms strive to achieve a stable electron configuration, often by gaining, losing, or sharing valence electrons to fill their outermost shell. This pursuit of stability drives chemical reactions.
Oxygen's Atomic Structure and Electron Configuration
Oxygen's atomic number is 8, which means a neutral oxygen atom possesses 8 protons and 8 electrons. To understand its valence electron count, we need to examine its electron configuration. Electrons occupy specific energy levels or shells around the nucleus. These shells have a maximum capacity for electrons: the first shell can hold up to 2 electrons, the second shell up to 8, and so on.
The electron configuration of oxygen is 1s²2s²2p⁴.
Let's break this down:
- 1s²: This indicates that the first energy level (n=1) contains 2 electrons in the 's' subshell.
- 2s²: The second energy level (n=2) has 2 electrons in the 's' subshell.
- 2p⁴: The second energy level also contains 4 electrons in the 'p' subshell.
The 's' and 'p' subshells are types of orbitals within each energy level that can accommodate different numbers of electrons. The 's' subshell can hold a maximum of 2 electrons, while the 'p' subshell can hold up to 6.
Determining Oxygen's Valence Electrons
Since valence electrons reside in the outermost shell, we look at the highest energy level in oxygen's electron configuration: the second energy level (n=2). This level contains a total of 6 electrons (2 from the 2s subshell and 4 from the 2p subshell).
Therefore, oxygen has 6 valence electrons.
The Significance of Six Valence Electrons: Oxygen's Reactivity
The presence of six valence electrons explains oxygen's high reactivity. Oxygen atoms strive to achieve a stable octet (8 electrons) in their outermost shell. They can achieve this in two primary ways:
1. Gaining Electrons: Ionic Bonding
Oxygen can gain two electrons to achieve a stable octet, forming an oxide ion (O²⁻). This process involves ionic bonding, where the oxygen atom attracts electrons from another atom with lower electronegativity, leading to an electrostatic attraction between the oppositely charged ions. For example, in the formation of magnesium oxide (MgO), magnesium loses two electrons to oxygen, resulting in Mg²⁺ and O²⁻ ions held together by ionic bonds.
2. Sharing Electrons: Covalent Bonding
Alternatively, oxygen can achieve a stable octet by sharing electrons with other atoms through covalent bonding. This involves the mutual sharing of electron pairs between atoms, creating a stable molecular structure. Oxygen is highly electronegative, meaning it strongly attracts electrons towards itself. This leads to the formation of polar covalent bonds, where the shared electrons are more closely associated with the oxygen atom. For example, in a water molecule (H₂O), oxygen shares two electron pairs with two hydrogen atoms, forming two covalent bonds and satisfying its need for eight valence electrons.
Oxygen's Role in Chemical Reactions and Biological Systems
Oxygen's reactivity due to its six valence electrons has profound consequences for chemical reactions and biological systems.
Combustion: A Vital Role
Oxygen's high reactivity is evident in combustion reactions. Oxygen acts as a strong oxidizing agent, readily accepting electrons from other substances, leading to the release of energy in the form of heat and light. This process fuels many essential industrial processes and is vital for energy production in many living organisms.
Respiration: Sustaining Life
In biological systems, oxygen plays a crucial role in cellular respiration, the process by which organisms convert glucose into ATP (adenosine triphosphate), the primary energy currency of cells. This process involves the reduction of oxygen to water, providing the energy needed for life's functions. The high electronegativity of oxygen makes it an effective electron acceptor in this process.
Oxidation and Reduction Reactions (Redox): A Central Player
Oxygen's tendency to gain electrons makes it a central player in redox reactions. These are chemical reactions that involve the transfer of electrons between species. Oxygen frequently acts as an oxidizing agent, accepting electrons and causing the oxidation of other substances. Conversely, in some reactions, oxygen can be reduced, gaining electrons from a reducing agent. These redox reactions are fundamental to many biological and industrial processes.
Oxygen's Importance and Conclusion
The seemingly simple question of how many valence electrons oxygen possesses unlocks a deeper understanding of its profound role in chemistry and biology. The six valence electrons dictate its high reactivity, driving its participation in numerous essential reactions, from combustion and respiration to countless other chemical processes. Understanding its atomic structure and the implications of its valence electron count is crucial for comprehending its importance in both natural and man-made systems. Its presence and reactivity are indispensable to life as we know it and underpin many technological advancements. The number 6, therefore, represents far more than just a simple atomic characteristic; it encapsulates the essence of oxygen's critical role in the universe.
Latest Posts
Latest Posts
-
Which Best Describes The Law Of Independent Assortment
Apr 06, 2025
-
You Should Always Assign The Needs Met Rating Before
Apr 06, 2025
-
Which Is The Most Densely Populated Continent
Apr 06, 2025
-
Average Velocity On Velocity Time Graph
Apr 06, 2025
-
The First Scientist To Observe Cells With A Microscope Was
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Valence Electrons Does Oxygen . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
