How Many Lone Pairs In Nh3
News Leon
Apr 05, 2025 · 5 min read
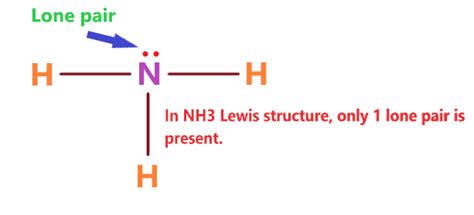
Table of Contents
How Many Lone Pairs in NH₃? A Deep Dive into Ammonia's Structure and Bonding
Ammonia (NH₃), a ubiquitous compound in nature and industry, presents a fascinating case study in molecular geometry and bonding. Understanding its structure is crucial to grasping its properties and reactivity. A key element of this understanding lies in determining the number of lone pairs of electrons on the nitrogen atom. This article will delve deep into the electronic structure of ammonia, explaining how to determine its lone pairs and exploring the implications of this feature on its various chemical and physical behaviors.
Understanding Lewis Structures and VSEPR Theory
Before we tackle the number of lone pairs in NH₃, let's establish a foundational understanding of two crucial concepts: Lewis structures and the Valence Shell Electron Pair Repulsion (VSEPR) theory.
Lewis Structures: A Visual Representation of Bonding
Lewis structures, also known as Lewis dot diagrams, are simplified representations of molecules that show the valence electrons of atoms and how they are shared in covalent bonds. They provide a visual way to understand the distribution of electrons within a molecule. In a Lewis structure, each bond is represented by a line (representing a shared pair of electrons), and lone pairs (unshared pairs of electrons) are depicted as dots.
VSEPR Theory: Predicting Molecular Geometry
The VSEPR theory helps us predict the three-dimensional arrangement of atoms in a molecule based on the repulsion between electron pairs in the valence shell of the central atom. Electron pairs, whether bonding or non-bonding (lone pairs), repel each other and try to get as far apart as possible. This repulsion dictates the molecule's geometry, influencing its properties.
Determining the Lone Pairs in NH₃: A Step-by-Step Approach
Now, let's apply these concepts to ammonia (NH₃) to determine the number of lone pairs on the nitrogen atom.
1. Determining Valence Electrons
- Nitrogen (N): Nitrogen is in group 15 of the periodic table, meaning it has 5 valence electrons.
- Hydrogen (H): Hydrogen is in group 1, meaning each hydrogen atom has 1 valence electron. Since there are three hydrogen atoms in NH₃, we have a total of 3 valence electrons from hydrogen.
Therefore, the total number of valence electrons in NH₃ is 5 (from N) + 3 (from 3H) = 8 valence electrons.
2. Constructing the Lewis Structure
- Central Atom: Nitrogen is the least electronegative atom and therefore acts as the central atom.
- Bonding Electrons: Each hydrogen atom forms a single covalent bond with the nitrogen atom, using 2 electrons per bond. This accounts for 6 electrons (3 bonds x 2 electrons/bond).
- Lone Pairs: The remaining valence electrons (8 total - 6 used in bonding = 2) form a lone pair on the nitrogen atom.
This gives us the Lewis structure for NH₃:
H
|
H - N - H
:
The two dots represent the lone pair of electrons on the nitrogen atom.
3. Applying VSEPR Theory
The nitrogen atom in NH₃ is surrounded by four electron pairs: three bonding pairs (to the three hydrogen atoms) and one lone pair. According to VSEPR theory, these four electron pairs will arrange themselves tetrahedrally to minimize repulsion. However, the molecular geometry (considering only the positions of the atoms) is trigonal pyramidal, not tetrahedral, because the lone pair is not considered when describing the molecular geometry.
Implications of the Lone Pair in Ammonia
The presence of the lone pair on the nitrogen atom significantly impacts ammonia's properties and reactivity.
1. Polarity
The lone pair contributes to the molecule's overall polarity. The nitrogen atom is more electronegative than the hydrogen atoms, and the asymmetrical distribution of electrons (due to the lone pair) creates a dipole moment, making ammonia a polar molecule. This polarity influences its solubility in polar solvents like water and its ability to participate in hydrogen bonding.
2. Hydrogen Bonding
The lone pair on the nitrogen atom allows ammonia to act as a hydrogen bond acceptor. The lone pair can attract a slightly positive hydrogen atom from another molecule, forming a hydrogen bond. This ability to participate in hydrogen bonding is responsible for ammonia's relatively high boiling point compared to other molecules of similar size.
3. Basicity
The lone pair makes ammonia a weak base. The lone pair can readily accept a proton (H⁺) from an acid, forming the ammonium ion (NH₄⁺). This is the basis of ammonia's behavior as a Brønsted-Lowry base.
4. Reactivity
The lone pair's availability influences ammonia's reactivity. It can act as a nucleophile, donating its electron pair to an electrophile, participating in various chemical reactions such as alkylation and acylation.
Comparing Ammonia with Other Molecules
To further solidify our understanding, let's compare the number of lone pairs in ammonia with other similar molecules.
Methane (CH₄)
Methane has a central carbon atom bonded to four hydrogen atoms. Carbon has four valence electrons, all of which are used in bonding with the hydrogen atoms. Therefore, methane has zero lone pairs. Its geometry is tetrahedral.
Water (H₂O)
Water has a central oxygen atom bonded to two hydrogen atoms. Oxygen has six valence electrons. Two electrons are used in each bond with hydrogen, leaving four electrons forming two lone pairs on the oxygen atom. Water, therefore, has two lone pairs. Its geometry is bent or V-shaped.
Hydrogen Fluoride (HF)
Hydrogen fluoride has a central fluorine atom bonded to one hydrogen atom. Fluorine has seven valence electrons. One electron is used in the bond with hydrogen, leaving six electrons forming three lone pairs on the fluorine atom. Thus, HF has three lone pairs.
Conclusion: The Significance of the Lone Pair in NH₃
In conclusion, ammonia (NH₃) has one lone pair of electrons on its nitrogen atom. This seemingly simple fact has profound implications for the molecule's properties and reactivity. The lone pair contributes to ammonia's polarity, hydrogen bonding capability, basicity, and nucleophilicity. Understanding the number and arrangement of lone pairs is essential for comprehending the behavior of ammonia and other molecules. By applying Lewis structures and VSEPR theory, we can accurately predict and interpret the molecular geometry and the resulting properties of molecules like ammonia, allowing us to understand their roles in various chemical and biological processes. Further investigation into related molecules helps solidify this understanding and provides a broader perspective on chemical bonding.
Latest Posts
Latest Posts
-
Are Ribosomes Made In The Nucleolus
Apr 06, 2025
-
The Total Energy Of A System Remains
Apr 06, 2025
-
An Effective Claim In An Argumentative Essay
Apr 06, 2025
-
Which Of The Following Statements Regarding Lipids Is True
Apr 06, 2025
-
Which Form Of Transport Requires The Expenditure Of Energy
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Lone Pairs In Nh3 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
