Good Conductor Of Heat And Electricity Metal Or Non-metal
News Leon
Apr 05, 2025 · 6 min read
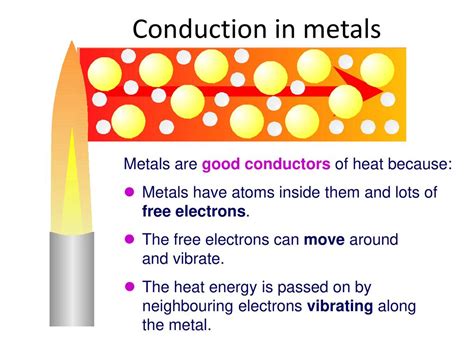
Table of Contents
Good Conductor of Heat and Electricity: Metal or Non-Metal?
The ability to conduct heat and electricity is a fundamental property of materials, crucial in numerous applications across various industries. From power transmission lines to cooking utensils, the choice of material hinges heavily on its conductivity. While both metals and non-metals exhibit varying degrees of conductivity, metals significantly outperform non-metals in this area. This article delves deep into the reasons behind this difference, exploring the atomic structure and electronic behavior that dictate conductivity. We'll also examine specific examples of excellent conductors and the unique characteristics that make them suitable for diverse applications.
The Atomic Structure: The Key to Conductivity
The key to understanding why metals are superior conductors lies in their atomic structure. Unlike non-metals, metals possess a unique arrangement of atoms and their electrons. This structure is characterized by:
1. Sea of Electrons:
Metal atoms have loosely bound valence electrons, not tightly associated with any particular atom. Instead, these electrons form a "sea" or "cloud" of delocalized electrons that can move freely throughout the entire metal structure. This "sea" is crucial for both heat and electrical conductivity.
2. Metallic Bonding:
The atoms in a metal are held together by metallic bonding, a type of chemical bond where the positively charged metal ions are embedded within this "sea" of delocalized electrons. The electrostatic attraction between the positive ions and the electron cloud provides the cohesive force holding the metal together. This flexible structure allows for easy electron movement.
3. Crystalline Structure:
Most metals exist in a crystalline structure, a highly ordered arrangement of atoms. This ordered arrangement facilitates the efficient flow of electrons and phonons (quanta of vibrational energy), contributing to high thermal and electrical conductivity.
Conductivity Mechanisms: A Closer Look
Let's examine the mechanisms behind the excellent conductivity of metals:
1. Electrical Conductivity:
Electrical conductivity is the ability of a material to allow the flow of electric current. In metals, the freely moving delocalized electrons respond readily to an applied electric field. The electric field exerts a force on these electrons, causing them to drift in a particular direction, thus constituting an electric current. The higher the density of these free electrons and the ease with which they can move, the higher the electrical conductivity.
2. Thermal Conductivity:
Thermal conductivity refers to the rate at which heat energy is transferred through a material. In metals, heat is primarily transferred through two mechanisms:
-
Electron Movement: The delocalized electrons, constantly in motion, efficiently transport kinetic energy (heat) throughout the material. When one part of the metal is heated, the energized electrons move to cooler regions, transferring their energy and thus equalizing the temperature.
-
Phonon Propagation: Lattice vibrations, or phonons, also contribute to heat transfer in metals. These vibrations propagate through the crystal lattice, transferring energy from hotter to cooler regions. However, in most metals, electron movement is the dominant mechanism for thermal conductivity.
Non-Metals: A Different Story
Non-metals, in contrast, exhibit significantly lower thermal and electrical conductivity. This difference stems from their atomic structure and bonding:
1. Localized Electrons:
Non-metal atoms hold their valence electrons tightly, forming covalent bonds with neighboring atoms. These electrons are localized, meaning they are not free to move throughout the material. This significantly restricts their ability to contribute to electrical or thermal conductivity.
2. Covalent and Ionic Bonding:
Non-metals primarily form covalent or ionic bonds. Covalent bonds involve the sharing of electrons between atoms, while ionic bonds involve the transfer of electrons from one atom to another, resulting in charged ions. Neither of these bond types leads to the formation of a "sea" of delocalized electrons like in metals.
3. Limited Electron Mobility:
The lack of free electrons and the strong bonding within non-metals severely limits the mobility of charge carriers, resulting in low electrical conductivity. Similarly, the restricted movement of electrons and the less efficient phonon propagation contribute to lower thermal conductivity.
Exceptions and nuances:
While the general trend is clear – metals are superior conductors – there are exceptions and nuances to consider.
-
Semiconductors: Semiconductors, such as silicon and germanium, are a class of materials with intermediate conductivity between metals and insulators. Their conductivity can be controlled by doping (introducing impurities) which alters their electronic properties and significantly improves their conductivity. This makes them extremely important in electronics.
-
Graphite: Graphite, a form of carbon, is an exception among non-metals. It possesses a layered structure, with delocalized electrons within each layer. This allows for relatively high electrical conductivity along the layers, although its conductivity is significantly lower than typical metals.
Examples of Excellent Metallic Conductors
Several metals stand out as exceptionally good conductors of heat and electricity. Their properties and applications vary, highlighting the versatility of metallic conductors:
1. Silver (Ag):
Silver boasts the highest electrical and thermal conductivity among all metals. Its superior conductivity makes it ideal for applications where minimal energy loss is crucial, such as high-frequency circuits and specialized electrical contacts. However, its high cost often limits its widespread use.
2. Copper (Cu):
Copper is a highly cost-effective alternative to silver, possessing excellent electrical and thermal conductivity. Its widespread use is evident in electrical wiring, plumbing, and heat exchangers. Its ductility and malleability make it easily workable for diverse applications.
3. Aluminum (Al):
Aluminum is lighter and less expensive than copper, making it a popular choice for high-voltage transmission lines and lightweight electrical components. Its high thermal conductivity also finds application in heat sinks and cookware.
4. Gold (Au):
Gold exhibits excellent corrosion resistance and conductivity, making it invaluable in electronic applications where long-term reliability is critical. It is frequently used in connectors and plating for electronic devices.
5. Tungsten (W):
Tungsten has a very high melting point and good electrical conductivity, making it ideal for use in incandescent light bulbs and high-temperature applications.
Applications: Harnessing the Power of Conductivity
The superior conductivity of metals drives a vast range of applications across various industries:
1. Electrical Power Transmission:
Copper and aluminum are the workhorses of electrical power transmission, forming the backbone of our electricity grid. Their high conductivity minimizes energy loss during long-distance transmission.
2. Electronics:
Metals like copper, gold, silver, and aluminum are essential in electronic devices, from integrated circuits to circuit boards, where their conductivity ensures efficient signal transmission and power distribution.
3. Heating and Cooling Systems:
Copper and aluminum tubes are widely used in heating and cooling systems, leveraging their high thermal conductivity for efficient heat transfer.
4. Cookware:
Copper and aluminum cookware benefit from high thermal conductivity, ensuring even heat distribution and faster cooking times.
5. Automotive Industry:
Aluminum's lightweight and high conductivity properties are increasingly utilized in automotive components such as wiring harnesses and heat sinks to improve fuel efficiency.
Conclusion: Metals Reign Supreme in Conductivity
The superior conductivity of metals compared to non-metals stems from their unique atomic structure, characterized by delocalized electrons and metallic bonding. This allows for efficient transport of both electrical charge and heat energy. While semiconductors represent an important exception with their controllable conductivity, metals remain the dominant choice for applications demanding high thermal and electrical conductivity. The specific metal chosen depends on factors like cost, durability, and specific application requirements. The continued development and refinement of metallic materials will undoubtedly shape future technological advancements, further highlighting the enduring importance of metallic conductivity.
Latest Posts
Latest Posts
-
Why Meiosis Is Called Reductional Division
Apr 06, 2025
-
What Is 150 Km In Miles
Apr 06, 2025
-
Which Of The Following Molecules Is Chiral
Apr 06, 2025
-
Ultimate Source Of Energy That Drives The Food Chain
Apr 06, 2025
-
Are Ribosomes Made In The Nucleolus
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Good Conductor Of Heat And Electricity Metal Or Non-metal . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
