Glucose Dissolves In Water Because It
News Leon
Apr 02, 2025 · 6 min read
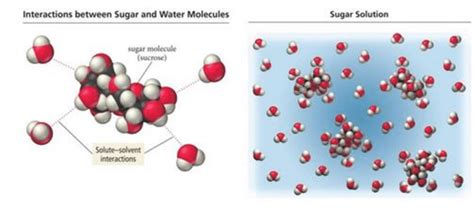
Table of Contents
Glucose Dissolves in Water Because… A Deep Dive into Solubility
Glucose, a simple sugar crucial for energy production in living organisms, readily dissolves in water. This seemingly simple observation belies a fascinating interplay of chemical forces and molecular properties. Understanding why glucose dissolves in water requires delving into the concepts of polarity, hydrogen bonding, and the thermodynamics of dissolution. This article explores these concepts in detail, offering a comprehensive explanation of glucose's solubility.
The Polar Nature of Glucose and Water
The key to understanding glucose's solubility lies in its molecular structure and the properties of water. Both glucose and water are polar molecules. This means they possess a slightly positive end and a slightly negative end due to an uneven distribution of electrical charge.
Understanding Polarity
Polarity arises from differences in electronegativity – the ability of an atom to attract electrons in a chemical bond. In water (H₂O), the oxygen atom is more electronegative than the hydrogen atoms. This creates a partial negative charge (δ-) on the oxygen and partial positive charges (δ+) on the hydrogens. This uneven charge distribution creates a dipole moment, making water a polar molecule.
Similarly, glucose (C₆H₁₂O₆) contains several hydroxyl groups (-OH). Oxygen is more electronegative than hydrogen, creating a partial negative charge on the oxygen and a partial positive charge on the hydrogen within each hydroxyl group. These polar hydroxyl groups contribute significantly to glucose's overall polarity. The presence of these polar groups makes glucose a polar molecule, capable of interacting with other polar molecules.
The Role of Hydrogen Bonding
The polarity of both glucose and water enables them to form hydrogen bonds. A hydrogen bond is a relatively strong type of intermolecular force that occurs between a hydrogen atom bonded to a highly electronegative atom (like oxygen or nitrogen) and another electronegative atom in a different molecule. These bonds are weaker than covalent bonds (which hold atoms together within a molecule), but they are significantly stronger than other intermolecular forces like van der Waals forces.
In the case of glucose dissolving in water, the hydroxyl groups of glucose form hydrogen bonds with the water molecules. The partially positive hydrogen atoms of water are attracted to the partially negative oxygen atoms of the glucose hydroxyl groups, and vice versa. This extensive network of hydrogen bonds between glucose and water molecules is the driving force behind glucose's solubility.
Thermodynamics of Dissolution: Enthalpy and Entropy
The dissolution of glucose in water is a process governed by thermodynamics. It involves two key factors: enthalpy (ΔH) and entropy (ΔS).
Enthalpy Change (ΔH)
Enthalpy refers to the heat content of a system. When glucose dissolves in water, energy is involved in breaking the existing intermolecular forces (hydrogen bonds) within both glucose and water and forming new hydrogen bonds between glucose and water.
The process of breaking the hydrogen bonds in both glucose and water requires energy input (endothermic process), leading to a positive enthalpy change. However, the formation of new hydrogen bonds between glucose and water releases energy (exothermic process), resulting in a negative enthalpy change. The overall enthalpy change (ΔH) for glucose dissolution is the net result of these two processes. While the exact value of ΔH can vary depending on the conditions, the overall process is generally slightly endothermic or close to zero, meaning the energy required to break the bonds is roughly balanced by the energy released when new bonds are formed.
Entropy Change (ΔS)
Entropy is a measure of disorder or randomness in a system. When glucose dissolves in water, the glucose molecules become dispersed throughout the water, increasing the overall disorder of the system. This increase in randomness contributes to a positive entropy change (ΔS). The increase in entropy is a significant driving force for the dissolution process.
Gibbs Free Energy (ΔG)
The spontaneity of a process is determined by the Gibbs free energy change (ΔG), which is related to enthalpy and entropy by the equation:
ΔG = ΔH - TΔS
where T is the temperature in Kelvin.
For glucose dissolving in water, the positive entropy change (ΔS) contributes significantly to a negative ΔG, even if the enthalpy change (ΔH) is slightly positive or close to zero. A negative ΔG indicates a spontaneous process, meaning that glucose will readily dissolve in water under normal conditions.
Factors Affecting Glucose Solubility
While glucose is highly soluble in water, several factors can influence its solubility:
-
Temperature: Generally, the solubility of glucose in water increases with increasing temperature. Higher temperatures provide more kinetic energy to the molecules, facilitating the breaking of bonds and increasing the rate of dissolution.
-
pH: The pH of the solution can influence glucose solubility, though the effect is relatively minor. Extreme pH values (very acidic or very basic) can potentially affect the glucose molecule's structure and thus its interaction with water, impacting its solubility.
-
Presence of other solutes: The presence of other dissolved substances in the water can affect glucose solubility through various intermolecular interactions. Some solutes might compete for hydrogen bonding with water, potentially reducing glucose's solubility.
Implications of Glucose Solubility
The high solubility of glucose in water is crucial for several biological processes:
-
Transport in the body: Glucose is readily transported in the bloodstream, allowing it to be delivered to cells throughout the body for energy production. Its solubility in water makes this transport efficient.
-
Cellular uptake: Glucose can easily pass through cell membranes, thanks to its solubility in the aqueous environment both inside and outside cells. Specific transport proteins also facilitate glucose uptake.
-
Metabolic pathways: Glucose participates in various metabolic pathways, including glycolysis and the Krebs cycle. Its solubility ensures its ready availability for these crucial biochemical reactions.
Comparison to Other Sugars
While glucose is highly soluble in water, the solubility of other sugars varies depending on their structure and the number of hydroxyl groups. For instance, fructose, another simple sugar, is even more soluble in water than glucose due to its slightly different molecular structure that allows for more extensive hydrogen bonding with water molecules. Larger, more complex carbohydrates like starch and cellulose have lower solubility in water due to their larger size and reduced number of exposed hydroxyl groups.
Conclusion: A Powerful Interplay of Forces
The high solubility of glucose in water is a direct consequence of its polar nature, the ability to form numerous hydrogen bonds with water molecules, and the favorable thermodynamics of the dissolution process. The positive entropy change, driven by the increased disorder upon dissolution, outweighs the small enthalpy cost of breaking existing intermolecular bonds. This fundamental understanding of glucose's solubility is critical in comprehending its vital role in biological systems and its widespread applications in various fields. Further research continues to refine our understanding of the intricacies of molecular interactions and their impact on solubility and other critical properties of chemical compounds. The seemingly simple process of glucose dissolving in water reveals a complex and fascinating interplay of forces at the molecular level.
Latest Posts
Latest Posts
-
Which Statement Best Describes The Law Of Conservation Of Energy
Apr 03, 2025
-
Find The Value Of I1 In The Figure
Apr 03, 2025
-
Which Is Closer To Earth Venus Or Mars
Apr 03, 2025
-
How Many Moles Are In One Liter
Apr 03, 2025
-
In What Organelle Does Respiration Occur
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Glucose Dissolves In Water Because It . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
