Ca Oh 2 Hcl Cacl2 H2o
News Leon
Apr 01, 2025 · 6 min read
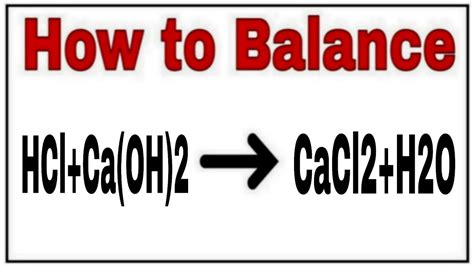
Table of Contents
The Reaction Between Calcium Hydroxide and Hydrochloric Acid: A Detailed Exploration of Ca(OH)₂ + 2HCl → CaCl₂ + 2H₂O
The reaction between calcium hydroxide, Ca(OH)₂, and hydrochloric acid, HCl, is a classic example of a neutralization reaction, a fundamental concept in chemistry. This reaction is highly exothermic, meaning it releases heat, and produces calcium chloride, CaCl₂, and water, H₂O. Understanding this reaction involves delving into the properties of each reactant, the mechanism of the reaction, its applications, and safety considerations. This comprehensive exploration will cover all these aspects.
Understanding the Reactants: Ca(OH)₂ and HCl
Before diving into the reaction itself, let's examine the individual properties of calcium hydroxide and hydrochloric acid.
Calcium Hydroxide: Ca(OH)₂
Calcium hydroxide, also known as slaked lime or hydrated lime, is an inorganic compound with the chemical formula Ca(OH)₂. It's a white, crystalline powder that is sparingly soluble in water, forming a solution known as limewater. Its key properties include:
- Strong Base: Ca(OH)₂ is a strong base, meaning it readily dissociates in water to release hydroxide ions (OH⁻), increasing the pH of the solution. This makes it highly alkaline.
- Alkaline Nature: Its alkaline nature makes it useful in various applications, from neutralizing acids to adjusting pH levels.
- Applications: It's used extensively in various industries, including construction (as a component of mortar and plaster), water treatment (for adjusting pH and softening water), and food processing (as a food additive).
Hydrochloric Acid: HCl
Hydrochloric acid, also known as muriatic acid, is a strong mineral acid with the chemical formula HCl. It's a colorless, highly corrosive liquid with a pungent odor. Key properties include:
- Strong Acid: HCl is a strong acid, meaning it readily dissociates in water to release hydrogen ions (H⁺), significantly lowering the pH of the solution. This makes it highly acidic.
- Highly Corrosive: Its corrosive nature necessitates careful handling and appropriate safety precautions.
- Applications: HCl has a wide range of industrial and laboratory applications. It’s crucial in the production of various chemicals, metal processing, and as a cleaning agent.
The Neutralization Reaction: Ca(OH)₂ + 2HCl → CaCl₂ + 2H₂O
The reaction between calcium hydroxide and hydrochloric acid is a classic example of an acid-base neutralization reaction. The strong acid (HCl) reacts with the strong base (Ca(OH)₂) to produce a salt (CaCl₂) and water (H₂O). The balanced chemical equation is:
Ca(OH)₂(aq) + 2HCl(aq) → CaCl₂(aq) + 2H₂O(l)
This equation shows that one mole of calcium hydroxide reacts with two moles of hydrochloric acid to produce one mole of calcium chloride and two moles of water. The "(aq)" indicates that the substance is dissolved in water (aqueous solution), while "(l)" denotes liquid water.
The Mechanism of the Reaction
The reaction proceeds through a simple proton transfer mechanism. The hydroxide ions (OH⁻) from calcium hydroxide accept protons (H⁺) from the hydrochloric acid. This process forms water molecules. Simultaneously, the calcium ions (Ca²⁺) and chloride ions (Cl⁻) remain in solution, forming calcium chloride.
OH⁻(aq) + H⁺(aq) → H₂O(l)
This fundamental acid-base reaction is driven by the strong electrostatic attraction between the oppositely charged ions.
Understanding the Products: CaCl₂ and H₂O
The products of the reaction are calcium chloride and water.
Calcium Chloride: CaCl₂
Calcium chloride (CaCl₂) is a white, crystalline solid that is highly soluble in water. It's a salt, meaning it's formed from the reaction of an acid and a base. Key properties and applications include:
- Hygroscopic: It readily absorbs moisture from the air, making it useful as a desiccant (drying agent).
- De-icing Agent: It's commonly used to melt ice and snow on roads and pavements.
- Food Additive: It's used as a food additive (E509) to firm food textures.
- Other Applications: It's employed in various industrial processes, including refrigeration and dust control.
Water: H₂O
Water (H₂O) is a ubiquitous substance crucial for life on Earth. Its properties as a solvent and its role in various chemical reactions are vital. In this reaction, the water formed is a byproduct of the neutralization process.
Applications of the Ca(OH)₂ + 2HCl Reaction
The reaction between calcium hydroxide and hydrochloric acid has several practical applications, including:
- pH Adjustment: This reaction is used to neutralize acidic solutions or to adjust the pH of a solution to a desired level. This is important in various industrial processes and in wastewater treatment.
- Chemical Synthesis: The reaction can be employed in the synthesis of calcium chloride, which, as noted above, has numerous industrial applications.
- Laboratory Experiments: It's a common experiment in chemistry education to illustrate the concept of neutralization reactions and stoichiometry.
- Wastewater Treatment: In wastewater treatment plants, Ca(OH)₂ can be used to neutralize acidic wastewater streams before discharge.
Safety Precautions
Both calcium hydroxide and hydrochloric acid are corrosive substances that require careful handling. Always follow these safety precautions:
- Wear appropriate Personal Protective Equipment (PPE): This includes safety goggles, gloves, and a lab coat.
- Work in a well-ventilated area: Hydrochloric acid releases fumes that can be irritating.
- Avoid direct contact: Avoid direct contact with both substances, as they can cause skin and eye irritation or burns.
- Neutralize spills carefully: If a spill occurs, neutralize it carefully with a suitable base (e.g., sodium bicarbonate) and follow appropriate cleanup procedures.
- Proper Disposal: Dispose of the waste products according to local regulations.
Further Considerations: Stoichiometry and Titration
Understanding the stoichiometry of the reaction—the molar ratios between reactants and products—is crucial for accurate calculations. This allows for determining the amount of calcium hydroxide required to completely neutralize a given amount of hydrochloric acid, and vice versa.
Titration is a quantitative analytical technique used to determine the concentration of an unknown solution (e.g., HCl) by reacting it with a solution of known concentration (e.g., Ca(OH)₂). This is often done using an indicator to visually determine the endpoint of the reaction.
Conclusion: A Fundamental Chemical Reaction with Broad Applications
The reaction between calcium hydroxide and hydrochloric acid is a simple yet fundamental chemical reaction with significant practical implications. Understanding the properties of the reactants, the mechanism of the reaction, and the characteristics of the products is essential for safe handling and application in various fields. The reaction's importance extends across numerous industries, from construction and food processing to wastewater treatment and chemical synthesis. Careful consideration of safety precautions is paramount when working with these chemicals. Further exploration into the stoichiometry and titration aspects offers a deeper understanding of the reaction's quantitative nature. This reaction serves as a cornerstone in chemical education and industrial processes alike.
Latest Posts
Related Post
Thank you for visiting our website which covers about Ca Oh 2 Hcl Cacl2 H2o . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
