Arrange The Following Amines In Order Of Decreasing Base Strength.
News Leon
Apr 05, 2025 · 6 min read
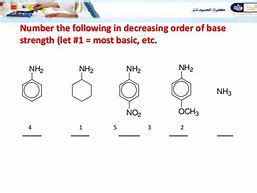
Table of Contents
Arranging Amines in Order of Decreasing Base Strength: A Comprehensive Guide
Amines, organic compounds derived from ammonia by replacing one or more hydrogen atoms with alkyl or aryl groups, exhibit varying degrees of basicity. Understanding the factors influencing their base strength is crucial in organic chemistry. This article provides a detailed explanation of the factors that determine amine basicity and guides you through the process of arranging amines in order of decreasing base strength. We will delve into the effects of alkyl groups, resonance, hybridization, and steric hindrance, providing numerous examples to solidify your understanding.
Understanding Amine Basicity
The basicity of an amine is determined by its ability to donate a lone pair of electrons to a proton (H⁺). A stronger base readily donates its lone pair, forming a stable conjugate acid. This ability is directly related to the availability and reactivity of the lone pair of electrons on the nitrogen atom.
Factors Affecting Amine Basicity:
Several factors intricately influence the basicity of amines. Let's explore each one in detail:
1. Inductive Effect of Alkyl Groups:
Alkyl groups are electron-donating groups (+I effect). They increase electron density on the nitrogen atom, making the lone pair more available for donation. Therefore, alkylamines are generally stronger bases than ammonia. The more alkyl groups attached to the nitrogen atom, the stronger the base. For instance, tertiary amines are stronger bases than secondary amines, which are stronger than primary amines, which in turn are stronger than ammonia.
- Example: (CH₃)₃N (trimethylamine) > (CH₃)₂NH (dimethylamine) > CH₃NH₂ (methylamine) > NH₃ (ammonia)
2. Resonance Effects:
Resonance significantly impacts amine basicity. If the lone pair on the nitrogen atom participates in resonance, it becomes less available for protonation, thus decreasing the base strength. Aromatic amines, such as aniline, are significantly weaker bases than aliphatic amines due to the resonance effect. The lone pair on the nitrogen atom in aniline is delocalized into the benzene ring, reducing its availability for protonation.
- Example: Aliphatic amines (e.g., methylamine) are stronger bases than aromatic amines (e.g., aniline).
3. Hybridization of Nitrogen:
The hybridization of the nitrogen atom affects the energy and availability of the lone pair. A nitrogen atom with sp³ hybridization has a lone pair in an sp³ orbital, which is less tightly held than a lone pair in an sp² orbital. Consequently, amines with sp³ hybridized nitrogen are generally stronger bases than those with sp² hybridized nitrogen.
- Example: Aliphatic amines (sp³ hybridized nitrogen) are stronger bases than amides (sp² hybridized nitrogen).
4. Steric Hindrance:
Steric hindrance refers to the hindrance caused by bulky groups around the nitrogen atom. Large groups surrounding the nitrogen atom can prevent the approach of a proton, hindering protonation and thereby decreasing the base strength. This effect is particularly noticeable in tertiary amines with bulky substituents.
- Example: Tertiary amines with bulky alkyl groups are weaker bases than tertiary amines with smaller alkyl groups, despite the +I effect of the alkyl groups. This is because the steric hindrance overshadows the inductive effect.
5. Solvation Effects:
The solvent in which the amine is dissolved plays a crucial role in determining its base strength. Solvation refers to the interaction between the solute (amine) and the solvent molecules. Amines that are better solvated are generally weaker bases because the solvent molecules stabilize the amine, making it less likely to donate its lone pair.
- Example: The relative basicity of amines can change depending on the solvent used. In protic solvents (e.g., water), solvation effects can significantly influence the observed base strength.
Arranging Amines in Order of Decreasing Base Strength: Practical Examples
Let's apply the knowledge gained to arrange several amines in decreasing order of base strength. We'll consider a variety of structures to illustrate the combined effect of the discussed factors.
Example 1:
Arrange the following amines in order of decreasing base strength:
- Triethylamine ((CH₃)₃N)
- Aniline (C₆H₅NH₂)
- Methylamine (CH₃NH₂)
- Ammonia (NH₃)
Solution:
The order of decreasing base strength is: Triethylamine > Methylamine > Ammonia > Aniline
- Triethylamine is the strongest base due to the +I effect of three methyl groups.
- Methylamine is stronger than ammonia due to the +I effect of one methyl group.
- Ammonia is a weaker base than methylamine due to the absence of an electron-donating alkyl group.
- Aniline is the weakest base because the lone pair on the nitrogen is delocalized into the benzene ring through resonance, making it less available for protonation.
Example 2:
Arrange the following amines in order of decreasing base strength:
- Diethylamine ((CH₃CH₂)₂NH)
- Diisopropylamine ((CH₃)₂CHNHCH(CH₃)₂)
- Ethylamine (CH₃CH₂NH₂)
- Pyrrole
Solution:
The order of decreasing base strength is: Diethylamine > Ethylamine > Diisopropylamine > Pyrrole
- Diethylamine is stronger than ethylamine due to the increased +I effect of two ethyl groups.
- Ethylamine is stronger than ammonia because of the +I effect of the ethyl group.
- Diisopropylamine, despite having two isopropyl groups (stronger +I effect compared to ethyl), is weaker than diethylamine due to the significant steric hindrance caused by the bulky isopropyl groups. This steric hindrance makes it difficult for a proton to approach the nitrogen atom.
- Pyrrole is the weakest base because the lone pair on the nitrogen is involved in the aromatic sextet, making it unavailable for protonation.
Example 3:
Arrange the following amines in order of decreasing base strength:
- Benzylamine (C₆H₅CH₂NH₂)
- p-Nitroaniline (NO₂C₆H₄NH₂)
- p-Methoxyaniline (CH₃OC₆H₄NH₂)
Solution:
The order of decreasing base strength is: Benzylamine > p-Methoxyaniline > p-Nitroaniline
- Benzylamine is the strongest base because the lone pair is not involved in resonance and the benzyl group has a weak +I effect.
- p-Methoxyaniline is stronger than p-nitroaniline because the methoxy group (-OCH₃) is an electron-donating group through resonance (+M effect), increasing the electron density on the nitrogen atom.
- p-Nitroaniline is the weakest base due to the presence of a strong electron-withdrawing nitro group (-NO₂) which decreases the electron density on the nitrogen atom through resonance (-M effect and -I effect).
Conclusion
Predicting the relative base strengths of amines requires a thorough understanding of the interplay between inductive effects, resonance, hybridization, steric hindrance, and solvation. By systematically analyzing these factors for each amine, you can accurately arrange them in order of decreasing base strength. Remember that the relative strengths may vary depending on the solvent used. This detailed guide equips you with the necessary knowledge to approach these problems confidently and accurately. The numerous examples provided serve as a valuable tool for practicing and solidifying your understanding of amine basicity. Further exploration of specific examples and their detailed analysis will enhance your mastery of this crucial concept in organic chemistry. Remember to always consider the combined effect of all factors when making comparisons.
Latest Posts
Latest Posts
-
What Is The Oxidation Number Of Iodine In Kio4
Apr 06, 2025
-
Which Of The Following Is Not True Concerning Crop Rotation
Apr 06, 2025
-
Abo Blood Types Are An Example Of A Mendelian Trait
Apr 06, 2025
-
Which Of The Following Is Most Acidic
Apr 06, 2025
-
What Is The Oxidation Number Of Cl In Hclo4
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Arrange The Following Amines In Order Of Decreasing Base Strength. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
