Are Combustion Reactions Endothermic Or Exothermic
News Leon
Apr 02, 2025 · 7 min read
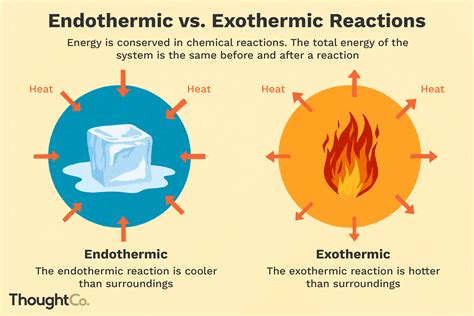
Table of Contents
Are Combustion Reactions Endothermic or Exothermic? A Deep Dive
Combustion reactions are a fundamental process in our daily lives, powering everything from our cars and power plants to our stoves and heating systems. Understanding the thermodynamics of combustion is crucial for various applications, from designing efficient engines to developing new fuels. The core question at the heart of this exploration is whether combustion reactions are endothermic or exothermic. The short answer is overwhelmingly exothermic. But let's delve deeper into the why, how, and implications of this fundamental characteristic.
Understanding Endothermic and Exothermic Reactions
Before diving into the specifics of combustion, let's define the key terms:
Endothermic Reactions: These reactions absorb energy from their surroundings. The energy of the products is higher than the energy of the reactants. You can often observe this as a decrease in temperature of the surroundings. Think of melting ice – it absorbs heat from the environment to change its state.
Exothermic Reactions: These reactions release energy into their surroundings. The energy of the products is lower than the energy of the reactants. This results in an increase in the temperature of the surroundings. A classic example is burning wood – it releases heat and light.
The Nature of Combustion Reactions
Combustion, at its simplest, is a rapid chemical reaction between a substance and an oxidant, usually oxygen, that produces heat and light. It's a type of redox reaction (reduction-oxidation), involving the transfer of electrons. The fuel (the substance being burned) is oxidized, while the oxidant (usually oxygen) is reduced.
The key characteristics of a combustion reaction include:
- Rapid reaction: The reaction occurs quickly, often explosively.
- Heat and light production: A significant amount of energy is released in the form of heat and light.
- Oxidant required: Oxygen is typically the oxidant, although other substances can act as oxidants under specific conditions.
- Production of oxidized products: The products of combustion are typically oxidized forms of the reactants. For example, the combustion of hydrocarbons produces carbon dioxide and water.
Why Combustion is Exothermic: A Look at Bond Energies
The exothermic nature of combustion stems from the differences in bond energies between the reactants and the products. Stronger bonds in the products compared to the reactants lead to a net release of energy.
Let's consider the combustion of methane (CH₄), a simple hydrocarbon:
CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(g)
In this reaction, the C-H bonds in methane and the O=O bonds in oxygen are broken. These bonds require energy input (endothermic process). However, the formation of C=O bonds in carbon dioxide and O-H bonds in water releases a significantly larger amount of energy (exothermic process). The net energy change is the difference between the energy required to break the bonds and the energy released when new bonds form. Since more energy is released than is absorbed, the overall reaction is exothermic.
This energy difference is the heat of combustion, a key measure of the energy released during a combustion reaction. It is typically expressed in kJ/mol (kilojoules per mole) and represents the amount of heat released when one mole of the fuel is completely burned.
Factors Affecting the Heat of Combustion
Several factors influence the heat of combustion:
- Type of fuel: Different fuels have different heats of combustion. For example, the heat of combustion of methane is higher than that of ethanol. This is because of variations in bond energies and molecular structures.
- Completeness of combustion: Complete combustion, where the fuel reacts completely with oxygen, produces the maximum amount of heat. Incomplete combustion, where there's insufficient oxygen, produces less heat and may generate harmful byproducts like carbon monoxide.
- State of matter: The state of matter (solid, liquid, or gas) of the fuel can affect its heat of combustion. Gaseous fuels generally have higher heats of combustion than liquid or solid fuels due to their higher energy states.
- Presence of other substances: The presence of other substances in the fuel or the environment can alter the heat of combustion. For example, additives in gasoline can slightly influence its energy output.
Applications of Combustion Reactions
The exothermic nature of combustion reactions has led to its widespread applications in various fields:
- Power generation: Combustion of fossil fuels (coal, oil, and natural gas) in power plants generates electricity.
- Transportation: Internal combustion engines in vehicles utilize the energy released from burning gasoline or diesel to propel vehicles.
- Heating: Combustion of natural gas or propane in furnaces and boilers provides heating for homes and industries.
- Industrial processes: Combustion is used in numerous industrial processes, such as cement manufacturing and metal refining.
- Cooking: Gas stoves and ovens utilize combustion to produce heat for cooking.
Environmental Implications
While combustion is essential for modern society, it also has significant environmental consequences. The burning of fossil fuels releases greenhouse gases, particularly carbon dioxide, contributing to climate change. Furthermore, incomplete combustion can produce harmful pollutants like carbon monoxide, particulate matter, and nitrogen oxides, contributing to air pollution and respiratory problems.
Exploring Different Types of Combustion
While the general principle holds true, the specifics of the exothermic nature can vary depending on the type of combustion:
Complete Combustion
This ideal scenario involves sufficient oxygen to fully oxidize the fuel, resulting in the maximum heat release and the production of primarily carbon dioxide and water. This maximizes the exothermic nature of the reaction. For hydrocarbons, the general equation is:
CₓHᵧ + (x + y/4)O₂ → xCO₂ + (y/2)H₂O + Heat
Incomplete Combustion
If the oxygen supply is limited, incomplete combustion occurs. This produces less heat and generates undesirable byproducts such as carbon monoxide (CO), soot (carbon particles), and other unburned hydrocarbons. The exothermic nature is reduced, and the overall process is less efficient.
Spontaneous Combustion
This occurs when a material ignites without an external ignition source. It typically results from slow oxidation processes that generate heat, accumulating until the ignition temperature is reached. The heat release is still exothermic, but the initiation is different.
Advanced Concepts and Further Considerations
Activation Energy
Even though combustion is exothermic (overall energy release), it still requires an initial input of energy, called activation energy, to initiate the reaction. This activation energy overcomes the initial energy barrier required to break the reactant bonds and start the chain reaction. Once the reaction starts, the energy released far surpasses the initial activation energy.
Thermodynamic Parameters
The exothermic nature of combustion can be quantified using thermodynamic parameters like enthalpy change (ΔH), which represents the heat change at constant pressure. For exothermic reactions, ΔH is negative, indicating a release of heat. Gibbs free energy (ΔG) further helps determine the spontaneity of the reaction. For combustion reactions, ΔG is usually negative, confirming their spontaneous nature under standard conditions.
Kinetics of Combustion
The speed of the combustion reaction is governed by its kinetics. Factors such as temperature, pressure, and surface area of the fuel influence the reaction rate. Higher temperatures and pressures generally accelerate combustion.
Alternative Fuels and Sustainable Combustion
The environmental concerns associated with fossil fuel combustion have fueled research into alternative fuels, such as biofuels and hydrogen. These fuels aim to reduce greenhouse gas emissions and improve sustainability. The combustion of these alternative fuels is still exothermic, but the overall environmental impact is significantly lessened.
Conclusion
In conclusion, combustion reactions are unequivocally exothermic. The energy released during these reactions is a result of the formation of stronger bonds in the products compared to the reactants. Understanding the exothermic nature of combustion is crucial for numerous applications, from energy production to industrial processes. However, it's equally important to address the environmental implications associated with combustion and strive for more sustainable practices and alternative fuels to minimize the negative effects on our planet. The continuous study and development in this field pave the way for cleaner and more efficient energy solutions.
Latest Posts
Latest Posts
-
A Mature Red Blood Cell Lacks A Nucleus Therefore It
Apr 03, 2025
-
Is Square Root Of 5 Irrational
Apr 03, 2025
-
Which Of The Following Is A Contact Force
Apr 03, 2025
-
What Is The Molar Mass Of Mg3 Po4 2
Apr 03, 2025
-
Rank The Compounds Below In Order Of Decreasing Base Strength
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Are Combustion Reactions Endothermic Or Exothermic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
