Activation Energy Of The Forward Reaction
News Leon
Apr 05, 2025 · 6 min read
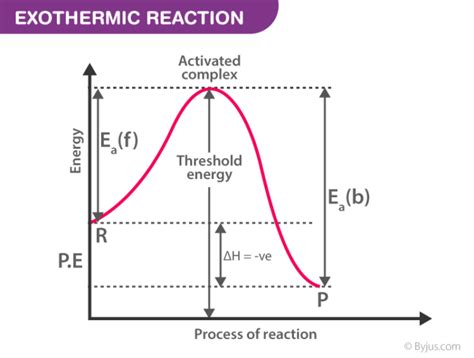
Table of Contents
Activation Energy of the Forward Reaction: A Deep Dive
Understanding chemical reactions and their rates is fundamental to many scientific fields. A key concept governing the speed of a reaction is the activation energy of the forward reaction. This article will delve into the intricacies of activation energy, exploring its definition, significance, factors influencing it, and its applications across various contexts.
What is Activation Energy?
The activation energy (Ea) of a forward reaction is the minimum amount of energy required for the reactant molecules to collide effectively and initiate a chemical reaction. It represents the energy barrier that must be overcome for reactants to transform into products. Think of it like pushing a boulder uphill – you need a certain amount of energy (activation energy) to get it over the crest (the transition state). Once it's over the crest, it rolls down the other side (forming products), releasing energy.
This isn't the overall energy change of the reaction (ΔH), which represents the difference in energy between reactants and products. Activation energy is specifically the energy needed to reach the transition state, an unstable, high-energy intermediate state between reactants and products.
The Role of Collisions
Chemical reactions occur when reactant molecules collide. However, not all collisions lead to a reaction. For a collision to be effective, it must possess sufficient energy to overcome the activation energy barrier and the correct orientation for bonds to break and form. Molecules with kinetic energy below the activation energy will simply bounce off each other without reacting.
Visualizing Activation Energy with Energy Diagrams
Activation energy is often depicted graphically using reaction energy diagrams (also known as reaction coordinate diagrams). These diagrams plot the potential energy of the system against the reaction coordinate (progress of the reaction). The diagram shows:
- Reactant energy: The initial energy of the reactants.
- Product energy: The final energy of the products.
- Activation energy (Ea): The difference in energy between the reactants and the transition state.
- ΔH (enthalpy change): The difference in energy between the reactants and the products. ΔH can be positive (endothermic reaction, absorbing heat) or negative (exothermic reaction, releasing heat).
These diagrams are crucial for visualizing the energy barrier and understanding the relationship between activation energy and reaction rate.
Factors Affecting Activation Energy
Several factors significantly influence the activation energy of a forward reaction:
1. Nature of Reactants:
The chemical nature of reactants plays a crucial role. Strong bonds require more energy to break than weaker bonds, leading to a higher activation energy. For example, reactions involving strong covalent bonds (like C-C or C-H) often have higher activation energies than those involving weaker bonds (like hydrogen bonds).
2. Reaction Mechanism:
The mechanism, or the step-by-step pathway of a reaction, significantly impacts activation energy. A reaction with a complex mechanism involving several intermediate steps usually has a higher activation energy than a reaction with a simple, one-step mechanism. Each step in the mechanism has its own activation energy, and the overall activation energy is determined by the highest energy barrier (the rate-determining step).
3. Temperature:
Temperature affects the kinetic energy of reactant molecules. At higher temperatures, molecules move faster and collide more frequently and with greater energy. This increases the proportion of molecules possessing sufficient energy to overcome the activation energy barrier, leading to a faster reaction rate. However, the activation energy itself doesn't change with temperature; only the fraction of molecules with enough energy to react increases.
4. Catalysts:
Catalysts are substances that increase the rate of a reaction without being consumed themselves. They achieve this by lowering the activation energy of the reaction. Catalysts provide an alternative reaction pathway with a lower energy barrier, allowing more reactant molecules to react at a given temperature. They do this by forming temporary bonds with reactants, facilitating bond breaking and formation. Enzymes are biological catalysts that lower activation energy in biological systems.
5. Surface Area:
For heterogeneous reactions (reactions involving reactants in different phases, such as a solid and a gas), the surface area of the solid reactant plays a role. A larger surface area provides more sites for reaction to occur, effectively increasing the frequency of effective collisions and lowering the apparent activation energy (though the true activation energy of the reaction itself remains unchanged).
6. Pressure:
In gas-phase reactions, increasing pressure increases the concentration of reactant molecules, leading to more frequent collisions. While this increases the reaction rate, it doesn't directly change the activation energy. The effect is similar to increasing the concentration of reactants in solution.
7. Concentration:
Higher concentrations of reactants lead to more frequent collisions, increasing the probability of successful collisions that overcome the activation energy barrier. Again, this increases the reaction rate but doesn't alter the activation energy itself.
Determining Activation Energy
The activation energy of a forward reaction can be determined experimentally using the Arrhenius equation:
k = A * exp(-Ea/RT)
where:
- k is the rate constant of the reaction
- A is the pre-exponential factor (frequency factor), representing the frequency of collisions with the correct orientation.
- Ea is the activation energy
- R is the ideal gas constant
- T is the temperature in Kelvin
By measuring the rate constant (k) at different temperatures, we can plot ln(k) against 1/T. The slope of the resulting straight line is equal to -Ea/R, allowing us to calculate Ea. This method is based on the fact that the Arrhenius equation can be linearized as:
ln(k) = ln(A) - Ea/RT
This linear form allows for easier graphical determination of Ea.
Significance and Applications of Activation Energy
Understanding activation energy is crucial in various fields:
1. Chemical Kinetics:
Activation energy is a fundamental parameter in chemical kinetics, governing the rate of chemical reactions. Predicting and controlling reaction rates is essential in various industrial processes, such as catalysis, polymerization, and combustion.
2. Catalysis:
The design and development of efficient catalysts rely heavily on understanding how catalysts lower activation energy. Researchers work to design catalysts with specific properties that minimize the activation energy barrier for desired reactions.
3. Biochemistry:
In biochemistry, activation energy is essential for understanding enzyme function. Enzymes act as biological catalysts, dramatically lowering the activation energy of biochemical reactions, allowing life processes to occur at a reasonable rate.
4. Materials Science:
Activation energy plays a significant role in material synthesis and processing. Understanding the activation energy of solid-state reactions is crucial for controlling the properties of materials, such as sintering, crystal growth, and phase transformations.
5. Environmental Science:
Activation energy is relevant to understanding the rates of environmental processes, such as atmospheric reactions, decomposition of pollutants, and geochemical transformations.
Conclusion
The activation energy of the forward reaction is a crucial concept in chemistry, providing insights into the rates and mechanisms of chemical reactions. Understanding its dependence on various factors allows for the prediction and control of reaction rates, which is vital in many scientific and industrial applications. From designing efficient catalysts to understanding biological processes, a grasp of activation energy is indispensable for advancements in numerous fields. Further research continuously refines our understanding of activation energy and its implications, driving innovation across diverse scientific disciplines.
Latest Posts
Latest Posts
-
Good Conductor Of Heat And Electricity
Apr 06, 2025
-
What Organelle Transports Proteins Around The Cell
Apr 06, 2025
-
Which Statements Are Correct Check All That Apply
Apr 06, 2025
-
Which Best Describes An Electrical Current
Apr 06, 2025
-
The Largest Endocrine Gland Is The
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Activation Energy Of The Forward Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
